
The role of inheritance in the development of adolescent varicoceles
Introduction
Varicoceles are the most common identifiable cause of male infertility, and is present in nearly 35% of patients with primary infertility. The frequency of varicoceles increases rapidly from 10 to 19 years, reaching a 15% prevalence by adulthood in the general population (1-3), and may increase by about 10% for each decade of life, reaching 75% by the eighth decade (4). However, its effect on paternity is less clear as 85% of males with varicocele were found to have fathered children without intervention in population studies (5-7). Although pathogenesis is poorly understood, varicoceles may alter testicular growth, spermatogenesis, and fertility potential in young adulthood (8,9). Therefore, early diagnosis of the pediatric varicocele is paramount (10,11).
Varicocele pathophysiology is thought to be multifactorial, with the primary factors believed to involve increased venous pressures in the left renal vein, valvular incompetence in the left internal spermatic vein, and a more acute angle at the junction with the left renal vein (12-16). Valvular incompetence is the primary anatomic abnormality associated with venous hypertension and is one of the primary factors behind the pathogenesis of varicose veins (13,14). Even with this shared pathophysiology, a population-based study looking at association between varicocele and varicose veins by Lai et al. was unrevealing, attributed to the unusually low prevalence of varicoceles in the populations (17).
Despite the high prevalence of varicoceles in the population and potential implications surrounding fertility, there are few studies examining the inheritance pattern in the general population, with none focusing specifically on an adolescent cohort. Mokhtari et al. and Gökçe et al. both described a significant increase in the prevalence of varicocele after a post-hoc examination of first-degree relatives (18,19). While genetic factors were also shown to make a strong contribution to the familial transmission of varicose veins from parents to offspring, no studies have looked into a possible hereditary association with varicocele (20). The current study had two primary aims: To calculate the prevalence of varicoceles in fathers of patients with diagnosed pediatric varicoceles, and additionally, to examine association between patients with varicocele and varicose veins in their first-degree relatives.
Methods
A series of 884 patients age 17 and above diagnosed with a varicocele between 2012 and 2015 based on ICD9 code 456.4 and ICD10 code 186.1 were identified. Families were then contacted, and permission obtained to discuss family history as was approved by our institutional review board. Each patient underwent a telephone survey, which inquired whether: (I) the patient’s father had varicoceles and/or the (II) patient’s father had varicose veins, (III) a sibling had varicoceles, and (IV) the patient’s mother had varicose veins prior to maternity, as well as whether or not family members who had positive histories underwent surgery. For the determination of the number of family members with a family history of varicocele and/or lower extremity varicose veins the father was considered to have had a positive history if he had varicoceles and/or varicose veins prior to achieving paternity.
Patient information include age at diagnosis, age at consent, grade of varicocele, and management [surgery or active surveillance (serial examinations and scrotal ultrasound)], as well as indication for surgery for those patients who underwent surgery. Descriptive statistics were performed in JMP®, Version 13, SAS Institute Inc., Cary, NC, 1989–2018. Logistic regression and Chi squared analysis were performed to assess differences between of the family history of select groups (e.g., grade 1, 2 or 3 varicoceles), and were performed in SPSS (IBM Corp. Released 2016, IBM SPSS Statistics for Mac, Version 24.0. Armonk, NY: IBM Corp).
Results
In total, 152 families (17.2%) responded with 139 families (91.4%) consenting to participate in the study. Among the patients with pediatric varicoceles (mean age 19.5±1.84 years old), the mean age at diagnosis was 14.5±2.5 years. In total 21.6% (30/139) of patients had surgery, 76.3% (106/139) were undergoing active surveillance, and 3 other individuals chose not to pursue any follow up. Among the 132 patients with data available, 6 (4.5%), 62 (47.0%), and 64 (48.5%) had varicocele grades of 1, 2, and 3 at the time of presentation, respectively. Active surveillance took place among all 6 patients with grade 1 varicoceles and 58 (93.5%) of the grade 2 patients. In contrast, 26 (40.6%) of the grade 3 patients had surgery.
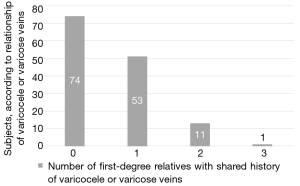
Overall, 65 (46.8%) patients had a family history of either varicoceles or varicose veins in first degree relatives. In total, 53 (38.1%) families had one first degree relative 11 (7.9%) had 2 affected first-degree relatives, and 1 (0.7%) patient had 3 affected first-degree relatives (Figure 1). Of the families with one first degree relative affected, 19 (13.7%) had a father who had varicocele and/or varicose veins, 11 (7.9%) had a sibling who had varicoceles, and 23 (16.5%) had a mother who had varicose veins (Table 1).

Full table
There were a total of 27 (19.4%) fathers with a history of either varicocele or varicose veins of which 16 (11.5%) had a history of a varicocele only, 9 (6.5%) had a history of varicose veins only, and 2 (1.4%) reported both varicocele and varicose veins. Of the 18 fathers who had a history of varicoceles, 10 (55.6%) had surgery. Eighteen (12.9%) of the patients had a sibling with a history of varicoceles (Table 2), with 1 (5.6%) requiring surgical intervention. Thirty-five (25.2%) mothers had a history of varicose veins of whom 3 (8.6%) had surgery.

Full table
Patients who required surgical management rather than active surveillance were not more likely to have a father with varicocele (P=0.274) or a mother with varicose veins (P=0.621). Patients with grade 2 and 3 varicoceles were more likely to have a father with a varicocele than those with a grade 1 varicocele (Table 3) on logistic regression analysis (P=0.037). Interestingly, patients with a grade 2 varicocele were more likely to have a father who required surgical ligation of a varicocele than grade 1 or grade 3 (P=0.019) on binary logistic regression (Table 3). Neither a sibling’s history of a varicocele (P=0.657), nor a mother’s history of varicose veins (P=0.306) was significantly associated with the grade of varicocele at diagnosis.

Full table
Discussion
To our knowledge, this is the first paper to document the prevalence of varicocele in fathers of adolescent patients with varicocele, as well as the first to look at a hereditary association between varicose veins in first-degree relatives and adolescents with varicocele. Given the results of this study, our findings suggest that while the prevalence of clinically significant varicoceles in fathers are equivalent to the general population, patients with higher grade varicoceles were more likely to have a father with varicocele than those with lower grade disease. It was also found that the father was more likely to require surgical ligation if the patient had grade 2 varicocele. This was not reflected in the siblings of patients. Our findings revealed no significant association existing between varicoceles in a patient and varicose veins in their parents.
In an early study conducted by Raman et al., the inheritance of varicoceles was evaluated by reviewing 44 patients with known varicocele and examining 62 of their first-degree relatives. Their results revealed 56.5% overall prevalence among first-degree relatives with 41% of fathers having palpable varicocele upon examination. This was significantly greater than their control population with a 6.8% prevalence (P<0.0001), which included men who presented for vasectomy reversal, all with proven fertility. Grade of varicocele was not predictive of inheritance in their findings (21). Critics of these findings explain this 8-fold significant difference by the control’s lower prevalence as not being representative of the general population (18,19).
Mokhtari et al. performed a prospective cohort study, in which the authors re-examined first-degree relatives of patients with known varicoceles and compared them to the scrotal exams of healthy kidney donor volunteers as a control (18). Overall, 45.4% of first-degree relatives were found to have a palpable varicocele compared to 11% in the control arm (P<0.001). The resulting prevalence among fathers was 26.6%. Gökçe et al. identified a 33.9% prevalence among first-degree relatives, compared to the control population prevalence of 12%. Among fathers 21.1% had varicocele at time of examination (19). In our study, the prevalence of varicoceles in fathers prior to achieving paternity were found to be comparable to that of the general population at 13%.
The present study counters the findings of Raman, Mokhtari, and Gökçe et al. These findings could be due to their wider range age of inclusion for first-degree relatives, as well as lower prevalence of varicocele in their control populations. Both Mokhtari and Gökçe et al. acknowledged the shared limitation of not including all first-degree relatives of the probands. In our study, the pediatric varicocele history of all fathers of the patients were obtained, allowing for a more complete determination of the inheritance implications among this cohort. Unfortunately, our study was unable to determine a complete first-degree relative prevalence without the inclusion of sibling prevalence. Neither Mokhtari, nor Gökçe et al. comment on the significance of varicocele grade, whereas the present study found significance among fathers in their increased likelihood of not only having varicocele, but also requiring surgical intervention when the patient had a varicocele of grade 2. While these findings were significant, it should be noted they may be due to an inadequate sample size as only 6 fathers had surgery.
The timing and necessity for varicocelectomy in men with pediatric varicoceles remains controversial. Evers et al. suggested in men with subfertility, varicocelectomy is not an effective treatment (22). However, Abdel-Meguid et al. reported improved semen in randomized controlled trials after varicocelectomy (23). Due to the potential implications on fertility, in the present study we focused on a younger cohort who had not yet achieved paternity; those whose paternity would most likely be impacted by clinical intervention. These 139 patients age of 17–22 years, were all diagnosed with pediatric varicocele (mean age at diagnosis 14.5±2.5 years). This is a contrast to Raman, Mokhtari, and Gökçe et al. studies, in which the patients’ age had a mean of 29.1 (±11.6, 14–77), 21.8 (±8.1, 17–34), and 27.4 (±5.9, 18–42) years respectively, none of which controlled for preconception (18,19,21).
A case-control population study performed by Lai et al. analyzed association between patients with varicocele and varicose veins in Taiwan (17). Their study included 2,727 patients with varicose veins and 10,908 controls in which they determined the prevalence of varicocele to be 1.3% among patients with varicoceles and 0.3% in the control population. The association between varicocele and varicose veins was determined to be significant (P<0.001). The low prevalence of varicocele in this study was believed to be underestimated, attributed to asymptomatic patients and lower rate of visiting doctors in Taiwan. Further, in a Swedish study examining the genetic contribution of varicose veins, adopted patients with at least 1 biologic parent diagnosed with varicose veins were more likely to develop varicose veins than adoptees without a biologic or adoptive parent with varicose veins, regardless of patient gender (20). Our results found no such association when looking at pediatric varicocele and varicose veins in first-degree relatives (P=0.306).
To our knowledge the present study represents not only the largest, but also the youngest cohort of primary patients with varicocele, and the only study to focus on pediatric varicoceles all of which had not achieved paternity. As such, this reveals a more accurate view of adolescent varicocele than prior publications, and denotes a population most likely to be affected by intervention. Our study also adds to the previous studies in an area not explored by the previous studies, with the findings of varicocele grade in the primary patient associated with surgical intervention in fathers.
The present study has several limitations. Without a control group, it is difficult to make comparisons and calculate true risks factors for varicoceles. Testing for associations was not performed for this study due to the small sample size and concerns regarding a potential source of bias. As the family histories were collected by phone interview and only included those families who chose to participate, the study may be subject to selection bias. There is also potential for recall bias as the family history data are self-reported. Further, any siblings of the proband may be under the age of 15, prior to puberty, when varicoceles are more likely to be observed.
Conclusions
While our data shows a father’s prevalence similar to that of the general population, the results of higher grade varicoceles associated with a higher prevalence of varicoceles and varicocele ligation, suggests the existence of a hereditary predisposition to high grade varicoceles. Furthermore, varicocele is not associated with varicose veins in first degree relatives. As this study contains results counter to previous reports, additional prospective studies are needed with a focus on younger adults with varicoceles who have not yet achieved paternity and their future offspring.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Ethics Committee of Cohen Children’s Medical Center (17-0521-CCMC) and written informed consent was obtained from all patients.
References
- Gorelick JI, Goldstein M. Loss of fertility in men with varicocele. Fertil Steril 1993;59:613-6. [Crossref] [PubMed]
- Witt MA, Lipshultz LI. Varicocele: a progressive or static lesion? Urology 1993;42:541-3. [Crossref] [PubMed]
- Kumanov P, Robeva RN, Tomova A. Adolescent varicocele: who is at risk? Pediatrics 2008;121:e53-7. [Crossref] [PubMed]
- Levinger U, Gornish M, Gat Y, et al. Is varicocele prevalence increasing with age? Andrologia 2007;39:77-80. [Crossref] [PubMed]
- Pinto KJ, Kroovand RL, Jarow JP. Varicocele related testicular atrophy and its predictive effect upon fertility. J Urol 1994;152:788-90. [Crossref] [PubMed]
- Safarinejad MR. Infertility among couples in a population-based study in Iran: prevalence and associated risk factors. Int J Androl 2008;31:303-14. [Crossref] [PubMed]
- Bogaert G, Orye C, De Win G. Pubertal screening and treatment for varicocele do not improve chance of paternity as adult. J Urol 2013;189:2298-303. [Crossref] [PubMed]
- Lipshultz LI, Corriere JN. Progressive testicular atrophy in the varicocele patient. J Urol 1977;117:175-6. [Crossref] [PubMed]
- Hienz HA, Voggenthaler J, Weissbach L. Histological findings in testes with varicocele during childhood and their therapeutic consequences. Eur J Pediatr 1980;133:139-46. [Crossref] [PubMed]
- Steeno O, Knops J, Declerck L, et al. Prevention of fertility disorders by detection and treatment of varicocele at school and college age. Andrologia 1976;8:47-53. [Crossref] [PubMed]
- WHO. The influence of varicocele on parameters of fertility in a large group of men presenting to infertility clinics. World Health Organization. Fertil Steril 1992;57:1289-93. [Crossref] [PubMed]
- Zampieri N, Cervellione RM. Varicocele in adolescents: a 6-year longitudinal and followup observational study. J Urol 2008;180:1653-6; discussion 1656. [Crossref] [PubMed]
- Takase S, Pascarella L, Bergan JJ, et al. Hypertension-induced venous valve remodeling. J Vasc Surg 2004;39:1329-34. [Crossref] [PubMed]
- Takase S, Pascarella L, Lerond L, et al. Venous hypertension, inflammation and valve remodeling. Eur J Vasc Endovasc Surg 2004;28:484-93. [Crossref] [PubMed]
- Stassen CM, Weil EH, Janevski BK. Left renal vein compression syndrome ("nutcracker phenomenon"). Rofo 1989;150:708-10. [Crossref] [PubMed]
- Gall H, Rudofsky G, Bähren W, et al. Intravascular pressure measurements and phlebography of the renal vein: a contribution to the etiology of varicocele. Urologe A 1987;26:325-30. [PubMed]
- Lai YW, Hsueh TY, Hu HY, et al. Varicocele is associated with varicose veins: A population-based case-control study. Int J Urol 2015;22:972-5. [Crossref] [PubMed]
- Mokhtari G, Pourreza F, Falahatkar S, et al. Comparison of prevalence of varicocele in first-degree relatives of patients with varicocele and male kidney donors. Urology 2008;71:666-8. [Crossref] [PubMed]
- Gökçe A, Davarci M, Yalçinkaya FR, et al. Hereditary behavior of varicocele. J Androl 2010;31:288-90. [Crossref] [PubMed]
- Kohno K, Niihara H, Li X, et al. Familial Transmission of Hospital-Treated Varicose Veins in Adoptees: A Swedish Family Study. J Am Coll Surg 2016;223:452-60. [Crossref] [PubMed]
- Raman JD, Walmsley K, Goldstein M. Inheritance of varicoceles. Urology 2005;65:1186-9. [Crossref] [PubMed]
- Evers JL, Collins JA. Assessment of efficacy of varicocele repair for male subfertility: a systematic review. Lancet 2003;361:1849-52. [Crossref] [PubMed]
- Abdel-Meguid TA, Al-Sayyad A, Tayib A, et al. Does varicocele repair improve male infertility? An evidence-based perspective from a randomized, controlled trial. Eur Urol 2011;59:455-61. [Crossref] [PubMed]


