Effects of varicocelectomy on serum testosterone
Introduction
Varicocele is the most common identified cause of male factor infertility (1). It is found in 15% of the general population, but is present in up to 35% of men presenting with primary infertility and up to 80% of men presenting with secondary infertility (1). The detrimental effect on spermatogenesis by varicocele is well documented, but not completely understood. It is has been proposed to be secondary to a variety of insults including oxidative stress, high scrotal temperature with loss of the scrotal counter-cooling mechanism, reflux of adrenal metabolites and gonadotropins or autoimmunity (2). Nonetheless, varicocelectomy has been shown to improve semen parameters in 70% of infertile men and approximately 40% will achieve pregnancy as a result (3). Given the effect of testosterone on spermatogenesis, it is hypothesized that low testosterone and Sertoli cell dysfunction may be intertwined (4,5). This paper reviews the current literature regarding varicocele and its effect on testosterone; both prior to and after repair.
Methods
Current literature in the Pubmed database was reviewed up to January 10th, 2016. The search terms “testosterone” and “varicocele” were used with a filter to the English language. A total of 337 articles were obtained. All abstracts were reviewed and twenty-eight studies examining the effect of varicocele on testosterone were included if they had pre and postoperative testosterone levels.
Review of literature
Much of the current literature regarding testosterone and varicoceles has been in the study of male infertility. There is little data examining varicocele repair for the sole purpose of treating low testosterone (6,7). Prior studies have demonstrated varicocele’s effect on Leydig cell dysfunction. Testicular biopsy in patients with idiopathic varicocele shows decreased tubular diameter, increased Leydig cell atrophy with vacuolization and decreased Leydig cells in testicular tissue when staining for testosterone (8). Venous pooling results in increased hypoxia, oxidative stress in seminiferous tubules and increased gonadotoxic renal and adrenal metabolites in the spermatic cord (9). Oxidative stress has been shown to correlate with varicocele grade and improves after subsequent repair (8). Furthermore, it has been previously demonstrated that as men age, serum testosterone levels decrease, predisposing men to sexual dysfunction; decreased libido, bone mineral density, strength, endurance, energy levels; sarcopenia; increased fatigue and depression; as well as impaired memory and concentration (10,11).
The majority of varicocele repairs are performed for male infertility or subfertility, with a smaller portion for associated pain or cosmetic reasons. It has been noted that varicocele is more common in men with secondary infertility compared to primary infertility suggesting a progressive decline in fertility associated with an untreated varicocele (12). When age matched to men with primary infertility, varicocele was attributed as the cause of secondary infertility in half of men (13). In a small study of 13 men with varicoceles, semen analysis was obtained at 9 and 96 months. No interventions were performed on any patients. At the 96-month interval, there was significantly decreased sperm density, motility and counts suggesting a progressive, deleterious effect (14). Varicocele repair has been shown to improve semen analysis and pregnancy rates (15,16), with the microsurgical approach having the lowest rate of varicocele recurrence and hydrocele formation (3). Percutaneous embolization is an alternative to surgery and technical success is achieved 75–90% of the time (17). However, the coils can migrate or refluxing veins missed, which can result in late recurrence or persistence after embolization (17,18).
Animal models
Recently, animal models have helped to better elucidate the relationship amongst varicocele, Leydig cell function and testosterone levels. Luo et al. utilized a rat model to compare a surgically induced varicocele with a sham operation that did not induce a varicocele (19). Intratesticular testosterone levels were obtained at four and eight weeks in both groups. There was noted to be Leydig cell apoptosis and diminished testosterone synthesis in the varicocele group at four and eight weeks and decreased intratesticular testosterone in the varicocele vs. the sham group at eight weeks (24.84 vs. 29.41 ng/g, P<0.05). Zheng et al. (20) demonstrated similar results, where 50 adolescent rats were divided into four groups: experimental varicocele (EV) without surgical correction, EV with artery-ligating varicocelectomy (EV + ALV), EV with artery-preserving varicocelectomy (EV + APV) and sham procedure control (no varicocele induced). Repair was performed six weeks after varicocele induction. Rats were reassessed six weeks after correction (twelve weeks after induction). Results are shown in Table 1. Intratesticular testosterone levels at twelve weeks were highest in the sham group followed by the EV + APV group. There was no statistical difference between the EV + APV group and sham control group, while the EV and EV + ALV resulted in decreased intratesticular testosterone levels.

Full table
Ozturk et al. (21) divided 21 rats into a sham control group, a varicocele induced group and a varicocele group with repair four weeks later. Intratesticular testosterone levels four weeks after repair (eight weeks after induction) were 13.62±4.83, 6.76±2.09 and 9.34±1.54 ng/g in the sham group, varicocele group and varicocele with repair group, respectively. There was statistical significance amongst all three groups. This demonstrated that four weeks after repair, intratesticular testosterone levels did improve with repair, although not to preoperative levels.
Although all three models demonstrate that varicocele results in decreased intratesticular testosterone that is subsequently alleviated with repair, the varicocele was induced acutely via surgical ligation and repaired within four to eight weeks. This acute presentation and quick repair rarely occurs in humans. However, the observed effect on intratesticular testosterone in such a short time period is notable. Nonetheless, these studies provide valuable insight into the effect of varicocele on testicular production of testosterone.
Introduction to varicocelectomy and serum testosterone change in humans
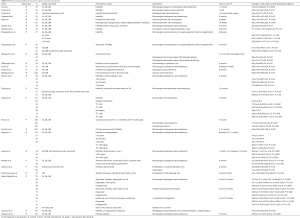
Twenty-eight studies (all shown in Table S1) were identified that had evaluated testosterone levels pre- and post-varicocele repair in men. The first study to suggest an effect of varicocele on testosterone was a retrospective study by Su et al. in 1995 (22), which showed total testosterone (TT) increase from 319±12 ng/dL preoperatively to 409±23 ng/dL (P<0.0004) postoperatively with inguinal or subinguinal microsurgical varicocelectomy in 33 infertile men. Cayan et al. demonstrated similar results (23). However, two retrospective studies in 2001 demonstrated no difference in 30 and 52 men undergoing retroperitoneal and microsurgical subinguinal varicocele repair for infertility, respectively (24,25). Initially, early studies demonstrated improvements in testosterone levels following varicocele repair (26-30), however, some more recent ones have failed to demonstrate this (6,7,31-36). A majority of the populations in studies without improvements in TT had normal TT preoperatively (7,24,31-36), possibly impairing the ability to detect changes in TT. Additionally, several of the studies without improved TT were designed to examine changes in SA and TT levels were a secondary outcome (7,32,33,35), therefore the studies may have lacked statistical power for detecting improvements. However, recent prospective studies examining the effect of varicocele repair in hypogonadal men appears to have resulted in a measured increase in TT. A recent meta-analysis of nine studies totaling 814 men reported a mean increase in TT of 97.48 ng/dL after surgical repair (37).
Full table
Varicocelectomy in hypogonadal men
Varicocele in the setting of low testosterone has been most extensively studied recently, with four studies of infertile men or subcohorts of men with low or low normal TT (38-41). Data is shown in Table 2.

Full table
In all four studies, hypogonadal men had improvements in TT following varicocele repair, the first of which was demonstrated in a prospective trial of 141 men undergoing infertility treatment (38). Eighty-three men underwent microsurgical varicocelectomy, while 38 men did not undergo repair. In the 49 men with TT <300 ng/dL, there was an increase from 219.3±65.8→358.1±94.0 ng/dL (P=0.0001) at six months postoperatively. There were also improved sperm count and motility in this group. There was no statistically significant increase in TT in men with TT >300 ng/dL in this same study. However, there were still improved sperm counts and motility in the repair group with TT >300 ng/dL. Interestingly, the control group of men with varicocele not undergoing surgical repair who were eugonadal did demonstrate a decrease in serum TT at 6 months from 499.5±134.8→465.7±127.0 ng/dL (P=0.006), albeit not to hypogonadal levels. There was not a change in SA in this subgroup.
Sathya Srini et al. looked at 200 men with infertility, clinical varicocele and TT <280 ng/dL. One hundred men underwent surgical repair and 100 men underwent assisted reproduction techniques (39). Those with surgical repair had an increase in TT of 177.2±18.44→301±43 ng/dL (P<0.001) at 6 months postoperatively. Those without repair had a TT decrease from 184.52±10.60→177±29 ng/dL (P=0.005) at 6 months. The results mirrored the semen analysis, where sperm count was observed to increase from 12.18±5.53→17.43±6.17 million/mL (P<0.001) and decrease, although not significantly, from 12.75±5.80→12.44±4.08 million/mL (P=0.683) in the intervention and control group, respectively.
Hsiao et al. retrospectively reviewed 78 men (82% for infertility) undergoing microsurgical subinguinal varicocele repair (40). All had TT <400 ng/dL. At an average of seven months postoperatively, there was a statistically significant increase in TT within the entire population from 308.4±7.1→417.5±14.8 ng/dL (P<0.0001) as well as men with TT <300 ng/dL and men with TT 301–400 ng/dL [248.7±8.6→357.2±25.0 ng/dL (P<0.0001) and 350.0±4.2→459±15.4 ng/dL (P<0.0001)]. There was no difference in TT improvements between the two groups.
Abdel-Meguid et al. recently prospectively analyzed 171 men with varicocele, infertility and abnormality on SA (41). Sixty-six infertile men with varicocele chose to undergo microsurgical subinguinal varicocele repair, 33 infertile men with varicocele chose assisted reproductive techniques, 33 fertile men (fertility definition not mentioned) with varicocele chose conservative management and 33 age-matched fertile men without varicocele were used as a control group. Twenty-eight men with TT <300 ng/dL demonstrated an increased TT level from 233.8±50.7→327.5±53.2→336.1±53.8 ng/dL at six and twelve months, respectively (P<0.0001) following varicocele repair. This is in contrast to 38 eugonadal men, who did not see any increase in TT with surgical intervention. Overall, all 66 men undergoing surgical repair did see an increase TT level [347.4±132.1→392±100.7→399±98.5 ng/dL at 6 and 12 months (P<0.0001)]. There was no difference between the initial TT levels in fertile vs. infertile men with varicocele. However, sperm counts were lower in the infertile men vs. fertile men with varicocele. The control group of fertile men without varicocele did have higher initial TT levels compared to all three other groups. There was no statistically significant change in TT in men undergoing assisted reproductive techniques or conservative management.
Although all of these four studies did demonstrate a benefit in TT with hypogonadal men, a retrospective study by Reşorlu et al. did not demonstrate any an increase in TT in a group of men who’s average starting TT was <300 ng/dL at 6 months postoperatively (6). TT levels did increase, but did not reach statistical significance. The study specifically was assessing if age affected improvements in semen analysis with TT changes as a secondary outcome. However, hypogonadism was not a specific inclusion criterion and the study was not designed for hypogonadal men. Additionally, there was no subgroup analysis of only hypogonadal men in the study.
Varicocele repair in hypogonadal men or low normal TT demonstrated improved TT in four studies. These results mirror prior literature demonstrating improved SA results in men with abnormal SA and varicocele, which suggests that varicocele, Sertoli cell and Leydig cell dysfunction may be intertwined. In addition to the above data, Ahmed et al. demonstrated improvement in TT in 81.8% of men with low TT compared to only 17.5% with normal TT (42). Interestingly, Zhody et al. (38) and Srini et al. (39) also noted that men who did not undergo repair had decreased TT. Abdel-Meguid et al. (41) did not observe these same results. All populations were small, but the data may suggest that not repairing varicocele in men results in developing or worsening hypogonadism. However, a larger population needs to be investigated to definitively reach this conclusion. Additionally, all men in these studies were infertile and it remains to be determined if these same effects can be seen in men presenting without infertility.
Varicocelectomy in older men
Most of the current literature studies testosterone and varicocele in men with infertility. Only two studies were identified (6,43) that examined age’s effect on TT and varicocele repair as shown in Table 3.

Full table
Reşorlu et al. retrospectively reviewed 96 men with infertility, scrotal swelling or pain undergoing microsurgical subinguinal varicocele repair (6). Their overall and subgroup analysis was subdivided into men 18–25, 26–35 and >36 years of age. They did not demonstrate any increase in serum TT levels at six months post operatively. Nor were there any differences in changes in TT amongst the age groups. Hsiao et al. reviewed 114 men with infertility or hypogonadism undergoing subinguinal microsurgical varicocele repair (42). Men were grouped as <30, 31–39 and >39 years of age. There were no statistically significant increases in TT in any group at an average of 10.1 months postoperatively. Nor were there any differences in changes in TT amongst the age groups. Although no improvement in TT was seen, both studies did show that in the ages studied, age did not have an effect on changes in TT. This data at least demonstrates that varicocele repair does not cause any adverse effects on TT in men in their late 30s–40s.
Varicocelectomy in men without infertility
There is limited data on men with low TT without infertility treated with varicocele repair. Data is shown in Table 4.

Full table
Reşorlu et al., as previously mentioned (6), reviewed 96 men with infertility, aesthetically displeasing scrotal swelling or scrotal content pain with varicocele. It was not reported what percentage were infertile. They did not demonstrate any statistically significant increase in TT with microsurgical subinguinal varicocelectomy. Rodriguez et al. studied 202 men who were referred for left testicular pain (76%) or left varicocele detected on employee health physical exam (7). No patients had a fertility complaint, yet those with proven fertility were not described. All men underwent inguinal varicocelectomy. Testosterone levels did increase from 648±156→709±232 ng/dL, however this was not significant. Sperm concentration and motility did increase 6 months postoperatively in men with normal sperm count or moderate oligozoospermia and in the overall population, respectively. There is very limited data on men undergoing repair without infertility. Although both populations did demonstrate improvements, neither was statistically significant. Rodriguez et al. is the largest population, however the starting TT was 648 ng/dL, raising the question whether increases in TT would provide benefit. Hypogonadal men without fertility complaints remain a population that would benefit most from further clinical study.
Subclinical varicocele repair
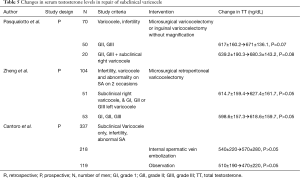
Subclinical varicocele was studied in two populations and data is shown in Table 5.
Full table
Pasqualotto et al. (32) and Zheng et al. (36) did not demonstrate improved TT when repairing subclinical right varicocele in addition to clinical left varicocele at 6 and 12 months postoperatively, respectively. Of note there was no difference in change in TT between men who only underwent left varicocele repair compared to those who also underwent subclinical right varicocele repair. Both populations starting TT was near 600 ng/dL raising the question whether improvement would be detected. This same study repeated in men with hypogonadism may demonstrate different results. Additionally, Cantoro et al. (44) prospectively analyzed 218 men with subclinical varicocele who underwent internal spermatic vein embolization compared to 119 men with subclinical varicocele refusing intervention. Although there was a rise in serum TT in the intervention group and a decrease in the observation group, neither was statistically significant at 6 months postoperatively. However, given that this population only underwent embolization rather than surgical repair, this data is difficult to interpret as no long term increases in TT have been shown from embolization apart from a single-center.
Varicocele treated with embolization
Embolization of varicocele is another treatment option. However, recurrence rates are higher and there is the risk of coil migration (17,18). Additionally, it relies on the expertise of an interventional radiologist to complete the procedure. In 2004, Gat et al. (26) retrospectively demonstrated improved TT with internal spermatic vein embolization in 83 men with infertility and varicocele. Six weeks post-procedure, their TT and FT increased from 12.07±6.07→17.22±8.43 nmol/L (P<0.001) and 5.93±2.44→10.21±7.69 nmol/L (P<0.001), respectively. Gat et al. (27) re-demonstrated this in a prospective trial of 101 with nonobstructive azoospermia or extremely severe oligoasthenoteratozoospermia (OAT) and varicocele. Six weeks after internal spermatic vein embolization, TT and FT levels increased from 9.81±5.67→18.23±8.09 nmol/L (P<0.001) and 5.10±2.37→10.59±5.13 nmol/L (P<0.001), respectively. However, other studies since then have been unable to confirm these results. Data is shown in Table 6.

Full table
Di Bisceglie et al. were unable to identify any improved TT in 38 men with infertility or local symptoms and varicocele that underwent spermatic vein sclerotherapy at six months post-procedure (33). Similarly, Cantoro et al. (44) and Prasivoravong et al. (45) did not demonstrate statistically significant improvements. However, Cantoro et al. embolized men with only subclinical varicocele, where TT demonstrated a non-statistically significant increase (44). This is comparison to 119 men used as control, who did not undergo intervention, but had a non-statistically significant decrease in TT. Prasivoravong et al. (45) prospectively analyzed 47 men with grade III varicocele, infertility and at least one abnormality on SA, who then underwent embolization of the left testicular vein. There was no significant change in serum TT after 3 and 6 months. Cantoro et al. (46) recently also demonstrated no improvement in TT in a retrospective study of 218 men with infertility or symptomatic varicocele undergoing retrograde internal spermatic vein scleroembolization after 6 months.
Despite initial promise that varicocele embolization results in improved TT (26,27), no studies apart from the initial single institution have reproduced this data (33,44-46). Currently it is difficult to recommend varicocele embolization to treat hypogonadism in men with varicocele. The microsurgical approach is the only approach that has persistently demonstrated improvement in TT for varicocele repair. The microsurgical approach allows for the most precise handling of tissue and avoidance of inadvertent lymphovascular injury that could otherwise contribute to further impaired testicular steroid dysfunction (47,48). As all studies that did show an increase in post-repair TT employed a microsurgical approach it would seem reasonable to use microsurgical techniques to provide the best outcome for treating low TT.
Discussion
Fifteen (22,23,26-30,38-43,49,50) of twenty-eight studies demonstrated an increase in TT following varicocele repair, typically at six to twelve months post repair. This was most prominently observed in hypogonadal men or men with low-normal TT. Of the fifteen studies with increased TT, twelve (22,26,27,30,38-43,49,50) were with average initial TT <400 ng/dL with six of seven prospective studies in hypogonadal or low-normal TT levels (27,38,39,41,42,49). When weighted according to patient population, the average starting TT for those with increased TT was 326 ng/dL while the average TT for those without increased TT was 535 ng/dL. This is a considerable difference between the two populations, which does raise the question whether population bias resulted in the increased TT. There was concern that only men with low or borderline low TT would have pre and postoperative data. However, the prospective studies demonstrated similar improvements in TT when looking at men with low or low-normal TT levels.
Ten of thirteen studies without improved TT had starting average TT >450 ng/dL (7,24,25,30,33,35,36,44-46). Therefore, given the already normal TT, it remains unlikely that an appreciable increase in serum TT would be observed. However, there has been no work performed to determine what period of time must pass before a varicocele results in hypogonadism or whether delaying repair adversely affects future testosterone levels. If the varicocele had not yet caused decreased testosterone producing capacity, it would be difficult to identify any increase in TT. There may be benefit in studying these populations now compared to men who presented with varicocele who did not undergo surgical repair as 10–15 years has passed.
To date, there have been no studies addressing the long-term effect of varicocele on testosterone production. While it has been previously shown that varicocele is not a static lesion when it comes to sperm production and that over time semen quality can decrease, it is also clear that not all men with varicocele become infertile. It would therefore be reasonable to conclude that not all men would necessarily become hypogonadal as a result of a varicocele. It should also be noted that in subgroup analysis, two of the studies did demonstrate a decrease in TT in men who elected not to undergo surgical repair (40,41). It could be hypothesized that men with varicocele are at greater risk for hypogonadism. However, it is possible that because these populations were infertile and had testicular dysfunction documented on SA, they were more susceptible to Leydig cell dysfunction. It remains to be seen if this can be translated to fertile men. Ultimately, a long-term study with a group of fertile men with varicocele who undergo surgical repair must be compared to men who decline surgical repair. This can help determine whether varicocele repair prevents future hypogonadism, the time period it develops and at what time point repair is indicated. Should repair only be undertaken when hypogonadism develops or when levels approach low-normal range? If repairing a varicocele in 30 years old can prevent hypogonadism and its effects in 60 years old, this would potentially reduce the development of hypogonadal symptoms at a later age and the cost of treatment.
Conclusions
Hypogonadism and varicocele do appear to be connected and repair results in elevated TT. This benefit appears greatest in hypogonadal or low-normal TT men, which has been demonstrated in multiple prospective control studies. After review of the current literature, microsurgical repair in a man presenting with hypogonadism and a varicocele appears to be a valid treatment option for appropriately counseled men. It has not been determined whether men with varicocele are at increased risk for developing low serum testosterone with clinical hypogonadal symptoms over time. Clearly, more research must be performed. Currently, the studies that did not demonstrate benefit in TT were in men with normal TT levels. This raises the question as to whether varicocele repair in the eugonadal state would reduce the likelihood of later hypogonadism.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Marc G. Surgical Management of Male Infertility. Campbells-Walsh Urology. 11th ed. Elsevier Publishing, 580-611.
- Choi WS, Kim SW. Current issues in varicocele management: a review. World J Mens Health 2013;31:12-20. [Crossref] [PubMed]
- Marmar JL, Agarwal A, Prabakaran S, et al. Reassessing the value of varicocelectomy as a treatment for male subfertility with a new meta-analysis. Fertil Steril 2007;88:639-48. [Crossref] [PubMed]
- Comhaire F, Vermeulen A. Plasma testosterone in patients with varicocele and sexual inadequacy. J Clin Endocrinol Metab 1975;40:824-9. [Crossref] [PubMed]
- Pirke KM, Vogt HJ, Sintermann R, et al. Testosterone in peripheral plasma, spermatic vein and in testicular tissue under basal conditions and after HCG- stimulation in patients with varicocele. Andrologia 1983;15:637-41. [Crossref] [PubMed]
- Reşorlu B, Cengiz K, Erhan S, et al. The Significance of Age on Success of Surgery for Patients with Varicocele. Int Urol Nephrol 2010;42:351-56. [Crossref] [PubMed]
- Rodriguez Peña M, Alescio L, Russell A, et al. Predictors of Improved Seminal Parameters and Fertility after Varicocele Repair in Young Adults. Andrologia 2009;41:277-81. [Crossref] [PubMed]
- Sirvent JJ, Bernat R, Navarro MA, et al. Leydig cell in idiopathic varicocele. Eur Urol 1990;17:257-61. [PubMed]
- Ito H, Fuse H, Minagawa H, et al. Internal spermatic vein prostaglandins in varicocele patients. Fertil Steril 1982;37:218-22. [Crossref] [PubMed]
- Orwoll E, Lambert LC, Marshall LM, et al. Testosterone and estradiol among older men. J Clin Endocrinol Metab 2006;91:1336-44. [Crossref] [PubMed]
- Harman SM, Metter EJ, Tobin JD, et al. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab 2001;86:724-31. [Crossref] [PubMed]
- Gorelick JI, Goldstein M. Loss of fertility in men with varicocele. Fertil Steril 1993;59:613-6. [Crossref] [PubMed]
- Witt MA, Lipshultz LI. Varicocele: a progressive or static lesion? Urology 1993;42:541-3. [Crossref] [PubMed]
- Chehval MJ, Purcell M. Deterioration of semen parameters over time in men with untreated varicocele: evidence of progressive testicular damage. Fertil Steril 1992;57:174-7. [Crossref] [PubMed]
- Yamamoto M, Hibi H, Hirata Y, et al. Effect of varicocelectomy on sperm parameters and pregnancy rate in patients with subclinical varicocele: a randomized prospective controlled study. J Urol 1996;155:1636-8. [Crossref] [PubMed]
- Kroese AC, de Lange NM, Collins J, et al. Surgery or Embolization for Varicoceles in Subfertile Men: Summary of a Cochrane Review. Fertil Steril 2014;102:1553-5. [Crossref]
- Nguyen HT, Etzell J, Turek PJ. Normal human ejaculatory duct anatomy: a study of cadaveric and surgical specimens. J Urol 1996;155:1639-42. [Crossref] [PubMed]
- Lurvey R, Durbin-Johnson B, Kurzrock EA. Adolescent varicocele: A large multicenter analysis of complications and recurrence in academic programs. J Pediatr Urol 2015;11:186.e1-6. [Crossref] [PubMed]
- Luo DY, Yang G, Liu J, et al. Effects of Varicocele on Testosterone, Apoptosis and Expression of StAR MRNA in Rat Leydig Cells. Asian J Androl 2011;13:287-91. [Crossref] [PubMed]
- Zheng YQ, Zhang XB, Zhou JQ, et al. The effects of artery-ligating and artery- preserving varicocelectomy on the ipsilateral testes in rates. Urology 2008;72:1179-84. [Crossref] [PubMed]
- Ozturk MI, Koca O, Keles MO, et al. The impact of unilateral experimental rat varicocele model on testicular histopathology, Leydig cell counts, and intratesticular testosterone levels of both testes. Urol J 2013;10:973-80. [PubMed]
- Su LM, Goldstein M, Schlegel PN. The effect of varicocelectomy on serum testosterone levels in infertile men with varicoceles. J Urol 1995;154:1752-5. [Crossref] [PubMed]
- Cayan S, Kadioglu A, Orhan I, et al. The effect of microsurgical varicocelectomy on serum follicle stimulating hormone, testosterone and free testosterone levels in infertile men with varicocele. BJU Int 1999;84:1046-9. [Crossref] [PubMed]
- Pierik FH, Abdesselam SA, Vreeburg JT, et al. Increased serum inhibin B levels after varicocele treatment. Clin Endocrinol (Oxf) 2001;54:775-80. [Crossref] [PubMed]
- Fujisawa M, Dobashi M, Yamasaki T, et al. Significance of serum inhibin B concentration for evaluating improvement in spermatogenesis after varicocelectomy. Hum Reprod 2001;16:1945-9. [Crossref] [PubMed]
- Gat Y, Gornish M, Belenky A, et al. Elevation of serum testosterone and free testosterone after embolization of the internal spermatic vein for the treatment of varicocele in infertile men. Hum Reprod 2004;19:2303-6. [Crossref] [PubMed]
- Gat Y, Bachar GN, Everaert K, et al. Induction of spermatogenesis in azoospermic men after internal spermatic vein embolization for the treatment of varicocele. Hum Reprod 2005;20:1013-7. [Crossref] [PubMed]
- Grober ED, Chan PT, Zini A, et al. Microsurgical treatment of persistent or recurrent varicocele. Fertil Steril 2004;82:718-22. [Crossref] [PubMed]
- Pasqualotto FF, Lucon AM, de Góes PM, et al. Relationship between the number of veins ligated in a varicocelectomy with testicular volume, hormonal levels and semen parameters outcome. J Assist Reprod Genet 2005;22:245-9. [Crossref] [PubMed]
- Ramasamy R, Schlegel PN. Microsurgical inguinal varicocelectomy with and without testicular delivery. Urology 2006;68:1323-6. [Crossref] [PubMed]
- Ishikawa T, Fujisawa M. Varicocele ligation on free testosterone levels in infertile men with varicocele. Arch Androl 2004;50:443-8. [Crossref] [PubMed]
- Pasqualotto FF, Lucon AM, de Góes PM, et al. Is it worthwhile to operate on subclinical right varicocele in patients with grade II-III varicocele in the left testicle? J Assist Reprod Genet 2005;22:227-31. [Crossref] [PubMed]
- Di Bisceglie C, Bertagna A, Baldi M, et al. Varicocele sclerotherapy improves serum inhibin B levels and seminal parameters. Int J Androl 2007;30:531-6. [Crossref] [PubMed]
- Lee RK, Li PS, Goldstein M. Simultaneous vasectomy and varicocelectomy: indications and technique. Urology 2007;70:362-5. [Crossref] [PubMed]
- Ozden C, Ozdal OL, Bulut S, et al. Effect of varicocelectomy on serum inhibin B levels in infertile patients with varicocele. Scand J Urol Nephrol 2008;42:441-3. [Crossref] [PubMed]
- Zheng YQ, Gao X, Li ZJ, et al. Efficacy of bilateral and left varicocelectomy in infertile men with left clinical and right subclinical varicoceles: a comparative study. Urology 2009;73:1236-40. [Crossref] [PubMed]
- Li F, Yue H, Yamaguchi K, et al. Effect of surgical repair on testosterone production in infertile men with varicocele: a meta-analysis. Int J Urol 2012;19:149-54. [Crossref] [PubMed]
- Zohdy W, Ghazi S, Arafa M. Impact of varicocelectomy on gonadal and erectile functions in men with hypogonadism and infertility. J Sex Med 2011;8:885-93. [Crossref] [PubMed]
- Sathya Srini V, Belur Veerachari S. Does varicocelectomy improve gonadal function in men with hypogonadism and infertility? Analysis of a prospective study. Int J Endocrinol 2011;2011:916380.
- Hsiao W, Rosoff JS, Pale JR, et al. Varicocelectomy is associated with increases in serum testosterone independent of clinical grade. Urology 2013;81:1213-7. [Crossref] [PubMed]
- Abdel-Meguid TA, Farsi HM, Al-Sayyad A, et al. Effects of varicocele on serum testosterone and changes of testosterone after varicocelectomy: a prospective controlled study. Urology 2014;84:1081-7. [Crossref] [PubMed]
- Ahmed AF, Abdel-Aziz AS, Maarouf AM, et al. Impact of varicocelectomy on premature ejaculation in varicocele patients. Andrologia 2015;47:276-81. [Crossref] [PubMed]
- Hsiao W, Rosoff JS, Pale JR, et al. Older age is associated with similar improvements in semen parameters and testosterone after subinguinal microsurgical varicocelectomy. J Urol 2011;185:620-5. [Crossref] [PubMed]
- Cantoro U, Polito M, Muzzonigro G. Reassessing the role of subclinical varicocele in infertile men with impaired semen quality: a prospective study. Urology 2015;85:826-30. [Crossref] [PubMed]
- Prasivoravong J, Marcelli F, Lemaître L, et al. Beneficial effects of varicocele embolization on semen parameters. Basic Clin Androl 2014;24:9. [Crossref] [PubMed]
- Cantoro U, Catanzariti F, Lacetera V, et al. Percentage change of FSH value: new variable to predict the seminal outcome after varicocelectomy. Andrologia 2015;47:412-6. [Crossref] [PubMed]
- Cayan S, Shavakhabov S, Kadioğlu A. Treatment of palpable varicocele in infertile men: a meta-analysis to define the best technique. J Androl 2009;30:33-40. [Crossref] [PubMed]
- Ding H, Tian J, Du W, et al. Open non-microsurgical, laparoscopic, or open microsurgical varicocelectomy for male infertility: a meta-analysis of randomized controlled trials. BJU Int 2012;110:1536-42. [Crossref] [PubMed]
- Shabana W, Teleb M, Dawod T, et al. Predictors of improvement in semen parameters after varicocelectomy for male subfertility: A prospective study. Can Urol Assoc J 2015;9:E579-82. [Crossref] [PubMed]
- Tanrikut C, Goldstein M, Rosoff JS, et al. Varicocele as a risk factor for androgen deficiency and effect of repair. BJU Int 2011;108:1480-4. [Crossref] [PubMed]


