An update on oxidized regenerated cellulose (fibrillar™) in reducing postoperative corporal bleeding following inflatable penile prosthesis surgery
Introduction
Inflatable penile prosthesis (IPP) is a well-established and increasingly popular treatment option for men with erectile dysfunction refractory to conservative pharmacological management. Advancements in device design and surgical technique over the last several decades have led to increased patient and partner satisfaction; accordingly, the number of IPP implantations rose by nearly 28% between 2000 and 2009 (1,2). As the number of men choosing IPP implantation continues to climb, so too does the incidence of postoperative complications include device infection, hematoma formation, urethral or corporal perforation, and glans erosion (3).
Development of a scrotal hematoma is a particularly serious complication of IPP surgery that exaggerates postoperative pain and swelling, prolongs convalescence, decreases patient satisfaction, and, most dangerously, escalates the risk of postoperative device infection due to bacterial seeding within the residual blood products. Various strategies have been employed to limit the incidence of postoperative hematoma formation and subsequent device infection, including “mummy wrap” compressive dressings (4), bedrest, sandbag compression, partial inflation of the cylinders (5-7), and closed suction scrotal drain placement (5,6,8,9). Even with these standard hemostatic practices, however, the incidence of scrotal hematoma development following IPP surgery remains fairly high at 0.5–2.9% (5,8-10).
Hemostatic agents have long been employed in orthopedic and neurosurgical procedures to limit hidden blood loss postoperatively (11,12), and they have been readily incorporated into various urological procedures (13). Similar agents, including collagen fleece grafts, have also been adopted within the sexual medicine community as a promising tool in urologic reconstructive surgery (14,15). However, there is scant data evaluating the utility of hemostatic agents specifically in urologic prosthetic surgery.
Oxidized regenerated cellulose (ORC) (Surgicel Fibrillar™) is a plant-based hemostatic agent. It is absorbable, loosely woven, and nicely suited for IPP surgery due to its pliability. Its acidic pH also offers bactericidal properties that extend to the skin flora commonly associated with prosthetic infections and to antibiotic-resistant organisms, providing an additional safeguard against postoperative device infection (16,17). Given the benefits hemostatic and antimicrobial activity could offer in urologic prosthetic surgery, we effected a practice change beginning in April 2016 wherein all men undergoing IPP implantation also had ORC pledgets placed bilaterally during corporotomy closure. We hypothesized that ORC placement within the corporal defects would result in reduced postoperative bleeding (measured in terms of 24-hour postoperative scrotal drain output) and published an initial paper in 2017 reporting our early positive experience with ORC in the first group of 32 men who underwent IPP implantation with ORC inclusion (18).
Since the time of our initial publication, we have continued to incorporate ORC into the corporotomy closures of all men undergoing IPP implantation. We hypothesized that this much larger collection of more comprehensive data would further and more definitively show that bilateral ORC placement during corporotomy closure in IPP surgery decreases postoperative scrotal drain output, a well-documented risk factor for development of a scrotal hematoma and associated complications.
Methods
After approval from the International Review Board, we reviewed data in a prospectively maintained IPP database to identify men who had an IPP placed or revised by a single surgeon between April 2013 and February 2019, excluding men who underwent concurrent artificial urinary sphincter (AUS) placement or concurrent penile plication. We began incorporating ORC pledgets during corporotomy closure in all IPP surgeries (implantations and revisions) in April 2016. All patients discontinued anticoagulation therapy 7 days prior to surgery and reinitiated treatment no sooner than 72 hours following surgery.
All prostheses (AMS 700 CX™, CXR™, and Coloplast TitanÒ) were placed using a trans-scrotal technique with high submuscular reservoir placement (19). During corporotomy closure, vascular forceps were used to place 2–3 ORC pledgets fashioned from a 1×2-inch Fibrillar™ package into both corporal defects, positioned to directly overlay the fully deflated IPP cylinders. The corporotomies were then closed over the absorbable ORC pledgets using a series of pre-placed 2-0 PDS traction sutures. A 9-mm suction drain was positioned across the closed corpora, and the remainder of the surgical wound was closed. The IPP cylinders were partially inflated and a compressive dressing with scrotal support was applied to further enhance hemostasis and to limit postoperative pain and swelling. The drain, compressive dressing, and a 14-F urethral catheter were left in place 24 hours postoperatively and removed either in the hospital or clinic setting on postoperative day 1, at which point cumulative scrotal drain output was documented.
Demographic, intraoperative, and postoperative data was retrospectively compared between the group of men who had ORC placed during corporotomy closure and the group of men who did not. We were interested in cumulative 24-hour postoperative scrotal drain output as our primary outcome. To secondarily assess scrotal hematoma-related complications, we also analyzed patient-initiated phone calls in the immediate postoperative period (up to 4 weeks following the surgery date). Complaints of scrotal pain/discomfort with associated bleeding, bruising, or swelling were documented.
Differences between the ORC and non-ORC cohorts were evaluated using the Mann-Whitney U test (continuous variables) and Fisher’s exact test (categorical variables). A P value less than 0.05 was considered to be statistically significant. All parameters that were discovered to be significantly associated with scrotal drain output on univariate analysis were then further evaluated using multivariable linear regression models.
Results
A total of 274 men underwent IPP insertion during the study period—175 (64%) in the ORC group and 99 (36%) in the non-ORC group. Patients who underwent concurrent AUS insertion or penile plication were excluded. The overwhelming majority of cases were first-time implants (n=232, 85%) rather than revision procedures; AMS 700 devices (n=182, 66%) also prevailed. 30% (n=82) of the patients were diabetic, 11% (n=31) had history of prior pelvic radiation, and 41% (n=111) were on anti-coagulation therapy. There were no significant differences in demographic or perioperative data among men with and without ORC (Table 1). While the predominant device brand did differ between the two groups (AMS 700CX™ in the ORC group versus Coloplast Titan® in the non-ORC group), no other significant device-related differences (i.e., total device length) were discovered between the ORC and non-ORC cohorts (Table 1).
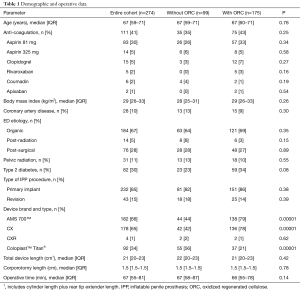
Full table
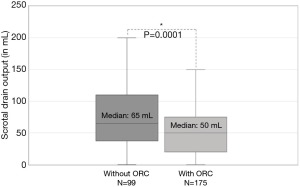
No complications associated with pledget positioning or increased intracorporal pressure (i.e., corporotomy rupture) were encountered. Median scrotal drain output was reduced by approximately 23% in the group of men who had ORC placed during corporotomy closure (50 vs. 65 mL; P=0.0001) (Figure 1). Linear regression analysis (Table 2) demonstrated that ORC usage was the only factor independently associated with reduced scrotal drain output (β: −32, 95% CI: −61 to −5; P=0.02). Secondary analysis of patient phone calls regarding scrotal complaints within the first 4 weeks following surgery showed both a reduction in the overall incidence of calls among men with ORC placement (42% vs. 55% of patients called the clinic postoperatively; P=0.04), as well as significantly fewer phone calls per patient (average 0.69 vs. 1.1; P=0.03) compared to the non-ORC group.
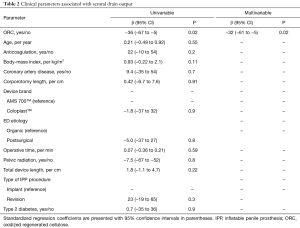
Full table
Nine men (3%) underwent subsequent IPP explantation during the study period, due to device infection in all cases. There were 5 explants in the ORC group (3%), compared to 4 explants (4%) in the non-ORC group (P=0.73). Incorporation of ORC during corporotomy closure thus did not present any increased risk for device infection or later explantation.
Discussion
In these 274 consecutive IPP implantations by a single surgeon, we demonstrate that placement of intracorporal ORC pledgets during corporotomy closure is an effective hemostatic strategy that reduces postoperative scrotal bleeding without heightening the risk for device infection or eventual device explantation. ORC usage was independently associated with reduced drain output, and secondarily, with fewer patient-initiated phone calls regarding scrotal complaints after surgery.
Hemostatic agents used in conjunction with standard hemostatic practices have repeatedly yielded promising results across a variety of surgical fields (13). Hemostatic agents have been readily incorporated into orthopedic procedures, proving to be particularly effective in reducing blood loss in knee and hip replacement operations (11,20). In neurosurgery, hemostatic agents have been very successful in managing capillary bleeding in tumor resection beds, as well as in controlling epidural oozing during spinal surgery (12). The primary urological interest has been in reducing bleeding during renal-sparing surgery, thus limiting the need for suture ligation of critical vessels (21-23). Yet, there is very little literature exploring the use of hemostatic agents in urologic prosthetic surgery. Delayed hemostasis leading to formation of a scrotal hematoma is a significant risk factor for device infection and failure, so the benefits that hemostatic agents could offer by promoting more efficient hemostasis and reducing the risk of bleeding-related complications is particularly important in urologic prosthetic surgery.
Fibrillar™ ORC is a natural, plant-based agent that enhances platelet adhesion and aggregation by acting as an absorbable lattice and mechanically activating both the intrinsic and extrinsic clotting cascades. We chose to utilize this hemostatic agent in particular because it is widely available and inexpensive, and its loosely knit cotton-like consistency allows for easy fashioning into small, firm pledgets that are ideal for insertion into the corporal defect during corporotomy closure (24). The product’s low pH also offers additional benefits in promoting local vasoconstriction and offering antimicrobial properties (12,17).
We observed a 23% reduction in postoperative drain output (≈15 mL) among the ORC cohort, though it does remain unclear if and how this reduction actually translates to a reduced risk for delayed scrotal hematoma development. As a proxy for scrotal complications, however, we also noted that ORC patients were significantly less likely to call the clinic complaining of scrotal pain or discomfort in the first 4 weeks following surgery. We hypothesize that any reduction in accumulation of residual blood products within the scrotum promotes convalescence, leading to enhanced recovery and increased patient satisfaction in the immediate postoperative period.
While we do acknowledge that the intracorporal positioning of the ORC pledgets only limits bleeding from the corporotomy site itself, we also believe hidden bleeding secondary to placement of the reservoir and/or scrotal dissection is unlikely to be clinically significant relative to bleeding from the corporotomy site.
Given that many patients traveled a significant distance to our institution for IPP surgery, variability in follow-up is certainly a limitation of this study that made defining the true incidence of scrotal hematoma formation a challenge. We employed cumulative scrotal drain output as a surrogate for hematoma development, but the clinical relevance of this parameter remains unclear. We did note that ORC patients initiated fewer phone calls regarding scrotal complaints in the immediate postoperative period, supporting our impression of enhanced recovery within this group, but additional research is indicated to elucidate all the factors contributing to postoperative patient phone calls. Further investigation is also warranted to evaluate if use of ORC is a cost efficacious tool in urologic prosthetic surgery, but ORC is fairly inexpensive (approximately $80 for the Fibrillar™ 1×2-inch packet) and it has been demonstrated to reduce overall healthcare costs in other surgical procedures and fields (25).
Given our low incidence of IPP explantation in both the ORC and non-ORC groups, we are unable to draw any definitive conclusions regarding ORC usage and rates of device infection. However, ORC use did not increase the incidence of device explantation in our study. Previous studies have also demonstrated that the low pH of ORC confers antibacterial properties that extend to both antibiotic-resistant microbes and to the skin flora most typically implicated in infection of prosthetic devices (17).
Conclusions
It is well-established that increased postoperative fluid accumulation within the scrotum is a significant risk factor for device infection following IPP implantation. By reducing postoperative drain output, ORC placement during corporotomy closure is an effective tool to potentially reduce scrotal hematoma-related complications in IPP surgery.
Acknowledgments
None.
Footnote
Conflicts of Interest: Dr. Allen F. Morey receives honoraria for being a guest lecturer/meeting participant for Boston Scientific and Coloplast Corp. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was approved by the Institutional Review Board at the University of Texas Southwestern Medical Center (IRB number: 102012-019).
References
- Fakhoury MQ, Halpern J, Bennett N. Recent advancements in penile prosthetics. F1000Res 2019. [Crossref] [PubMed]
- Montague DK. Penile prosthesis implantation in the era of medical treatment for erectile dysfunction. Urol Clin North Am 2011;38:217-25. [Crossref] [PubMed]
- Krzastek SC, Smith R. An update on the best approaches to prevent complications in penile prosthesis recipients. Ther Adv Urol 2019;11:1756287218818076. [Crossref] [PubMed]
- Henry GD, Wilson SK. Updates in inflatable penile prostheses. Urol Clin North Am 2007;34:535-47. [Crossref] [PubMed]
- Wilson SK, Cleves M, Delk JI. Scrotal Hematoma Prevention Following Penile Prosthesis Implantation: To Drain or Not to Drain. J Urol 1996;55:634A.
- Köhler TS, Hellstrom WJ. Using wound drains for the uncomplicated penile prosthesis. J Urol 2011;186:1187-9. [Crossref] [PubMed]
- Antonini G, Busetto GM, De Berardinis E, et al. Minimally invasive infrapubic inflatable penile prosthesis implant for erectile dysfunction: evaluation of efficacy, satisfaction profile and complications. Int J Impot Res 2016;28:4-8. [Crossref] [PubMed]
- Kramer A, Goldmark E, Greenfield J. Is a closed-suction drain advantageous for penile implant surgery? The debate continues. J Sex Med 2011;8:601-6. [Crossref] [PubMed]
- Sadeghi-Nejad H, Ilbeigi P, Wilson SK, et al. Multi-institutional outcome study on the efficacy of closed-suction drainage of the scrotum in three-piece inflatable penile prosthesis surgery. Int J Impot Res 2005;17:535-8. [Crossref] [PubMed]
- Garber BB, Bickell M. Delayed postoperative hematoma formation after inflatable penile prosthesis implantation. J Sex Med 2015;12:265-9. [Crossref] [PubMed]
- Wang JQ, Chen LY, Jiang BJ, et al. Oxidized Regenerated Cellulose Can Reduce Hidden Blood Loss after Total Hip Arthroplasty: A Retrospective Study. J Invest Surg 2019;32:716-22. [PubMed]
- Keshavarzi S, MacDougall M, Lulic D, et al. Clinical experience with the surgicel family of absorbable hemostats (oxidized regenerated cellulose) in neurosurgical applications: a review. Wounds 2013;25:160-7. [PubMed]
- Hong YM, Loughlin KR. The use of hemostatic agents and sealants in urology. J Urol 2006;176:2367-74. [Crossref] [PubMed]
- Hatzichristodoulou G, Gschwend JE, Lahme S. Surgical therapy of Peyronie’s disease by partial plaque excision and grafting with collagen fleece: feasibility study of a new technique. Int J Impot Res 2013;25:183-7. [Crossref] [PubMed]
- Hatzichristodoulou G. Evolution of the surgical sealing patch TachoSil® in Peyronie’s disease reconstructive surgery: technique and contemporary literature review. World J Urol 2019. [Epub ahead of print]. [Crossref] [PubMed]
- Achneck HE, Sileshi B, Jamiolkowski RM, et al. A comprehensive review of topical hemostatic agents: efficacy and recommendations for use. Ann Surg 2010;251:217-28. [Crossref] [PubMed]
- Spangler D, Rothenburger S, Nguyen K, et al. In vitro antimicrobial activity of oxidized regenerated cellulose against antibiotic-resistant microorganisms. Surg Infect (Larchmt) 2003;4:255-62. [Crossref] [PubMed]
- Rozanski AT, Viers BR, Liu AG, et al. Oxidized Regenerated Cellulose (Fibrillar) Reduces Risk of Postoperative Corporal Bleeding Following Inflatable Penile Prosthesis Surgery. Urology 2017;108:190-4. [Crossref] [PubMed]
- Morey AF, Cefalu CA, Hudak SJ. High submuscular placement of urologic prosthetic balloons and reservoirs via transscrotal approach. J Sex Med 2013;10:603-10. [Crossref] [PubMed]
- Saleh A, Hebeish M, Farias-Kovac M, et al. Use of Hemostatic Agents in Hip and Knee Arthroplasty: A Critical Analysis Review. JBJS Rev 2014. [Crossref] [PubMed]
- Ploussard G, Haddad R, Loutochin O, et al. A combination of hemostatic agents may safely replace deep medullary suture during laparoscopic partial nephrectomy in a pig model. J Urol 2015;193:318-24. [Crossref] [PubMed]
- Kommu SS, McArthur R, Emara AM, et al. Current Status of Hemostatic Agents and Sealants in Urological Surgical Practice. Rev Urol 2015;17:150-9. [PubMed]
- Sundaram CP, Keenan AC. Evolution of hemostatic agents in surgical practice. Indian J Urol 2010;26:374-8. [Crossref] [PubMed]
- Cohen SD, Francois Eid J. Hemostatic Matrix During Corporotomy Closure. J Sex Med 2014;11:869-72. [Crossref] [PubMed]
- Martyn D, Kocharian R, Lim S, et al. Reduction in hospital costs and resource consumption associated with the use of advanced topical hemostats during inpatient procedures. J Med Econ 2015;18:474-81. [Crossref] [PubMed]