
Comparison and outcomes of nonobstructive azoospermia patients with different etiology undergoing MicroTESE and ICSI treatments
Introduction
Nonobstructive azoospermia (NOA), which is identified in about 15% of infertile men, is the most severe manifestation of male infertility (1). Due to an intrinsic testicular impairment of sperm production, the success rate of conventional sperm retrieval [open testicular biopsy and testicular fine needle aspiration (FNA)] remains very low (2). The option of most men of NOA was limited to donor insemination, although minority of them may have the opportunity of intracytoplasmic sperm injection (ICSI) treatment using rare sperm from their own testes.
With the advances of microsurgery, a significant higher sperm retrieval rate (SRR) was obtained compared to conventional procedures (3). During microdissection testicular sperm extraction (MicroTESE), with the use of an operating microscope, it is possible to identify and selectively extract larger seminiferous tubules which have higher probability of harboring spermatozoa. MicroTESE has the advantage of minimal invasion, safety and limited disruption of testicular function, with low risks of postoperative intratesticular bleeding and decrease in the serum testosterone concentration (4). Potential predictive significance of clinical and laboratory findings for SRR has been debated in various studies. Prognostic factors have included testis volume, serum follicle-stimulating hormone (FSH), inhibin B, genetic alterations and histological testicular pattern (5-7), however, some studies showed contrary opinions that none of these parameters could precisely predict MicroTESE outcome solely (4).
The present study aimed to compare clinical, laboratory, histological features and MicroTESE outcomes of NOA of various etiologies, so to identify the determinant factors for MicroTESE outcomes in NOA. In some cases, cryopreservation of testicular sperm is necessary when sperm still remains after fresh sperm-oocyte-ICSI treatment or when ICSI could not be arranged on the same day of sperm retrieval operation. The argument between the use of fresh or frozen-thawed spermatozoa from MicroTESE needs further research. In this study, we also investigate ICSI outcomes using fresh and frozen thawed sperms. The information based on our study would be useful in counseling the patient and his partner in choosing the appropriate assisted reproductive techniques treatment and procedure.
Methods
Patient selection
In the present study, we retrospectively studied 595 men with NOA who underwent MicroTESE in an attempt to find sperms for ICSI over a 5-year period from January 2013 to December 2017 in reproductive centers in Ren Ji Hospital, Shanghai and Northwest Women’s and Children’s Hospital, Xian, Shaanxi Province. Given the fact that MicroTESE is an invasive procedure, our criteria for classifying testicular failure is strict, which included semen analysis results, history and physical examinations, endocrine profiles, and genetic testing. Additionally, we confirmed the diagnosis by histological evaluation. The detailed diagnosis of NOA was established based on the following data: (I) at least three different centrifuged ejaculates, 5 min at 3,000 rpm centrifugation and screening at 400× magnification using an inverted microscope, according to the World Health Organization guidelines (8). (II) Other thorough evaluations, including history, physical examination, hormone profile [serum FSH, luteinizing hormone (LH) and total testosterone (T)], scrotal ultrasound, genetic testing (Yq microdeletions and karyotyping) were available for all patients. The testis volume was measured using scrotal ultrasound. All the patients had their serum hormone levels measured using chemiluminescence immunoassay technique, with normal values in the range: 1.5–14 mIU/mL for FSH, 1.5–8 mIU/mL for LH, 9.4–37 nmol/L for total testosterone. Giemsa Karyotype was used for standard cytogenetic procedure in all cases by analysis of at least 20 G-banded metaphases from a peripheral blood lymphocyte culture, and in all cytogenetically normal cases. Molecular screening for Yq microdeletions was carried out on DNA extracted from peripheral blood using PCR. (III) Testicular failure was confirmed by TESE or FNA prior to or during operation, so to exclude obstructive cause. Histological patterns of NOA include the following histopathological patterns: (I) presence of Sertoli cells only syndrome (SCO); (II) maturation arrest, incomplete spermatogenesis, not beyond the spermatocyte stage; and (III) hypospermatogenesis, all cell types up to spermatozoa are present, but there is a distinct decline in the number of reproducing spermatogonia (9). Common etiology conditions associated with NOA include genetic abnormalities [e.g., Y chromosome microdeletions (YCMDs) and Klinefelter syndrome (KS), congenital abnormalities (e.g., cryptorchidism), post-infectious (e.g., mumps orchitis), exposure to gonadotoxins (e.g., chemotherapy, radiotherapy), and idiopathic. Obstructive azoospermia, anejaculation, patients with complete AZFa or AZFb microdeletions, abnormal karyotyping such as 46,XX, chromosome translocation, were excluded.
Surgical procedure
MicroTESE was performed under general anesthesia described by Schlegel (10). The procedure began on the right one if there was no difference between the two testicles or the one with obviously larger volume. After a midline incision is made in the scrotum, the testis is delivered and the tunica vaginal is opened. Testicular parenchyma is observed directly at ×15–20 magnification to locate and collect the wider seminiferous tubule with higher chance of harboring spermatozoa. The procedure is terminated when spermatozoa are retrieved or when further dissection may likely jeopardize the testicular blood supply (11). Successful retrievals were defined as the detection of sperm. A fragment of testicular parenchyma was fixed in Bouin’s solution for histopathological examination simultaneously.
Sperm processing and freezing
Testicular fragments were washed in Earl’s Balanced Salt Solution (EBSS Sigma, USA) medium to remove blood, then they were placed in sterile tissue culture dishes (Falcon, Franklin Lakes, NJ, USA) with 0.5 mL of Sperm Washing Medium™ (from Vitrolife, Sweden), and finely minced using sterile hypodermic injection needles. They were directly examined under an inverted microscope at ×200 magnifications for the presence of spermatozoa. The results were regarded as positive when at least one recoverable sperm was observed. If sperm is not found on the day of surgery, the search continues on the next day morning on the remaining cell suspensions. If oocyte collection is not arranged the same day or the next day of MicroTESE, sperm was routinely cryopreserved. Testicular tissues were mixed 1:1 with sperm freezing medium (Origio, Denmark) in 0.5 mL straw (Croy Bio System, France) after labeling stayed 30 min in liquid nitrogen vapor before being stored in liquid nitrogen. Thawing was performed at 37 °C for 5 min. The preparation was then separated from the cryoprotectant by washing in culture medium and centrifugation at 2,000 rpm for 15 min. The resulting pellet was resuspended in 500 µL of culture medium (Vitrolife, Sweden). If we collected oocytes on the same day, fresh testicular sperm samples were used after centrifuging for 15 min at 2,000 rpm.
Embryo evaluation and transfer
Briefly, fertilization was considered to have occurred after the visualization of the two pro-nuclei stage of the oocyte 24 hours after the intracytoplasmic injection of the motile spermatozoa. Embryo cleavage was checked approximately 48 and 72 hours after ICSI. The embryos were classified as top quality when they had three to four symmetrical blastomeres on the second day of culture and seven to eight symmetrical blastomeres on the third day. On the third day after oocyte pick-up, embryos were transferred transcervically into the uterine cavity under ultrasound guidance. Pregnancy was determined as a spontaneous rise in a β-hCG concentration on day 10 after embryo transfer. Clinical Pregnancy was confirmed by visualization of an intrauterine gestational sac showing cardiac activity under ultrasonographic examination at week 6–7.
Statistical analysis
Statistical analysis was performed using the SPSS 12.0 software. Results are expressed as means ± SD for numeric variables. Categorical variables were expressed as proportions (%). Differences between groups of patients in medians for quantitative variables and differences in distributions for categorical variables were tested with the Kruskal-Wallis on way analysis of variance (ANOVA) and Chi-square test, respectively. P values less than 0.05 were considered statistically significant.
Results
A total of 595 NOA patients received MicroTESE in our study. Patient’s average age was 29.4±6.2 years (range of 23–48 years). FSH, LH and T serum levels of patients were 21.04±11.36 IU/L, 9.68±5.87 IU/L and 12.55±5.59 nmol/L respectively. Mean right testicular volume was 7.8±3.6 mL, mean left testicular volume was 7.6±3.3 mL. Fifty-three patients reported the history of sperm detection in the ejaculate or partner pregnancy. Two patients reported that their partners had the history of early pregnancy, but aborted at the first 2 months of pregnancy. The other 51 patients have rare sperm detection in the past, but no sperm has been detected in the last three semen analysis. A proportion of 35.8% (19/53) of them have positive detestation, but no higher than those who have no history of sperm detection or pregnancy (40.8%, 221/542). As to the etiology, 48 patients were idiopathic, 2 with AZFc microdeletion, 2 have cryptorchidism history, 1 with KS. Histopathological examination showed that 71.09% (423/595) of NOA patients were SCO, 23.03% (137/595) of them were maturation arrest, 5.88% (35/595) were hypospermatogenesis.
Patients’ demographic characteristics, baseline endocrine profile, testicular volume and histopathology results from various causes are presented in Table 1. Etiology of NOA was defined for 595 patients: 66 (11.1%) were KS, 34 (5.7%) with microdeletion of the AZFc, 33 (5.5%) cases had the history of bilateral cryptorchidism (all underwent orchiopexy before adulthood), 13 (2.2%) had a history of mumps orchitis, 3 (0.5%) underwent cancer-relative chemotherapy, and the rest 446 (75.0%) were idiopathic NOA. There was no significant difference in the mean age of NOA patients between each group. Testicular volume in KS group is much smaller compared to the other groups (P<0.05). Serum FSH and LH of KS group is significantly higher compared with that of other groups (P<0.05). There were no significant differences in testicular volume, serum FSH, LH and T levels between the other five groups. The most majority pathological type of each group was SCO except NOA of mumps orchitis and chemotherapy, in which groups ratio of maturation arrest and SCO were quite similar.
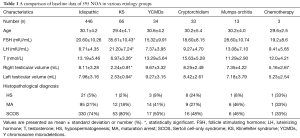
Full table
Our results showed that the overall SRR was 40.3% (240/595). But not all the retrieved testicular sperms were suitable for the following ICSI treatment. According to the criteria of embryo laboratory, only normal morphology spermatozoa could be used for ICSI. Among those successfully retrieval cases, 83.8% (201/240) of them had their sperms used in subsequent ICSI treatment, the rest 39 cases gave up further ICSI treatment either due to severely abnormal morphology of spermatozoa, or to the spermatozoa remaining completely immotile even after culture. SRR of the cryptorchidism (84.8%, 28/33) and mumps (84.6%, 11/13) groups were much higher than that of other groups, the SRR of idiopathic group was the lowest (31.8%, 142/446), SRR and the percentage of sperm qualified for ICSI of each group were presented in Table 2. SRR of different testicular histopathology was as the follows: SCO group was 37.1% (157/423), maturation arrest group was 39.4% (54/137), hypospermatogenesis group was 82.9% (29/35).
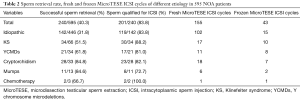
Full table
We used fresh sperm in 155 and frozen sperm in 38 NOA patients. One hundred and ninety-eight ICSI cycles utilizing MicroTESE sperm were retrospectively analyzed, including 155 fresh MicroTESE ICSI cycles and 43 frozen-thawed MicroTESE ICSI cycles. The reasons for sperm freezing were as following: (I) sperm retrieval and oocyte obtaining could not be arranged on the same day for various causes; (II) the female partner would accept ICSI treatment by husband’s sperm, but refuse to ICSI by donor’s sperm; (III) failure to obtain mature oocyte, so ICSI could not be arranged simultaneously.
Neither the age of male nor that of their partner showed significant differences between the two groups. Fertilization rate, cleavage rate, and clinical pregnancy rate of fresh sperm group were slightly higher than those in frozen thawed MicroTESE sperm group, but high qualified embryo rate of fresh sperm group was lower than frozen thawed group. The differences were of no statistical meaning (see Table 3).
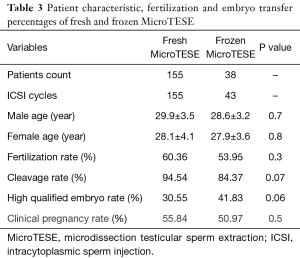
Full table
Discussion
NOA is an untreatable testicular failure resulting from various causes, which may be classified as primary or secondary. Primary disorders include genetic causes such as microdeletions in Y-chromosome and KS, or congenital origins such as cryptorchidism. Secondary causes may be due to orchitis, chemo-or radiotherapy, trauma, or endocrine dysfunction. It has been shown that mature spermatozoa can be found in the testes in part of NOA patients despite the absence of ejaculated spermatozoa in their semen because of the existence of isolated foci of active spermatogenesis. Studies have shown that only more than 3 mature spermatids exist per seminiferous tubule’ cross-section that spermatozoa can be detected in ejaculate. Men with NOA have a mean of 0–3 mature spermatids per seminiferous tubule, that is why rare sperms are occasionally found in ejaculates (12,13).
MicroTESE with subsequent ICSI treatment is now the only option for men with NOA to achieve biological offspring. However, given the financial burdens and risks relative to oocyte retrieval, it would be beneficial to be able to predict the success of sperm retrieval using non-invasive parameters before the procedure. Nevertheless, active spermatogenesis regions could not be predicted in general, for example, FSH may reflect the predominant pattern of spermatogenesis. Inhibin B is found to be slightly more sensitive than FSH as an index of spermatogenic status, however, FSH or inhibin B alone or in combination fail to predict the successful outcome of TESE for patients with NOA (14-16). Studies to date have shown that no one single factor has been found to correlate with successful sperm retrieval absolutely.
NOA of definite etiology, such as cryptorchidism, has relatively high SRR compared to idiopathic NOA. In our study, the SRR of cryptorchidism group (84.8%) ranked the highest among all the groups, which is in accordance with others’ reports (17-19). Orchiopexy has no benefit for seminiferous tubules undergoing irreversible degeneration, but it helps preserve the foci of germ cells of normal spermatogenesis. Negri et al. compared 30 bilateral cryptorchid men of NOA with 77 men with various causes of non-obstructive azoospermia and concluded that bilateral cryptorchid was a positive predictive factor for sperm finding after TESE (20). In a study of 38 azoospermia men who had previously undergone orchidopexy, correlations were found between sperm recovery rate and age at orchidopexy and testicular volume. Men who had had surgery before ten years old had a better sperm recovery rate than those who had had orchidopexy older than ten years of age. But Wiser et al. studied 40 patients of NOA who had undergone orchidopexy and found no statistically significant SRR differences between men who had undergone surgery before or after ten years of age, and no significant relationship between sperm recovery rate and testicular volume (21).
Sperm were successfully retrieved in 51.5% of KS group in our study, which was slightly lower than that of some studies (22). A possible explanation is that rare non-disjunction events may select for XY clones in developing germ cells, which are then able to proceed through meiosis to form normal haploid spermatogonia. Men with NOA lose some focal areas of spermatogenesis and may have less spermatozoa maturation with aging. This is important for patient counseling when urologists attempt a MicroTESE in men with small testes.
In our study, idiopathic NOA accounts for 75.5% of all NOA patients, the SRR of this cohort was relatively low (31.8%) compared to other groups. There is a high probability that idiopathic NOA is caused by genetic defects, such as gene mutation, gene polymorphism and epigenetic alterations, which lead to spermatogenic failure directly or indirectly depending on environmental factors, epigenetic modification, etc. In addition to acquired causes, the well-established genetic screening currently are only Karyotype and Y-chromosome microdeletions. Partial deletion of Y-chromosome, DNA methylation, testis-specific genes mutation may be the real causes of abnormality of spermatogenesis or meiosis (23-25).
When sperm collection was not possible on the day of oocyte retrieval for ICSI in some cases, sperm cryopreservation in advance is preferred and has been shown to be feasible. Cryopreservation of testicular spermatozoa from MicroTESE for future ICSI cycles would avoid both unnecessary female stimulation and repetitive injuries for successive ICSI cycles (26). Several authors have demonstrated that ICSI treatments with fresh or frozen testicular spermatozoa have similar results (27,28). Consider Tavukcuoglu’s and Karacan’s reports (29,30), which involved comparison of fertilization rate, embryo transfer, embryo quality, pregnancy rate and birth rate of fresh and frozen thawed MicroTESE sperm, no significant differences were observed between the two groups. Our study also showed that freezing does not affect fertilization rate, embryo transfer, pregnancy rate. High qualified embryo rate of fresh sperm group was slightly lower than that of frozen thawed group. It may be related to freezing itself, which is also a method of screening sperm. Sperm of poor quality is eliminated by freezing-thaw procedure. The similar results above don’t mean that patients can choose either procedure randomly. There exists dilemma when too few sperms are acquired; the rare sperm may be lost during the thawing procedure. Using fresh sperm for ICSI would be an appropriate choice in such condition.
Since we haven’t the ability to prognose the outcome of sperm retrieval or oocyte retrieval accurately, it is difficult to choose a perfect solution when developing an IVF program for patients undergoing MicroTESE. If NOA patients are more likely to have sperm, such as patients with bilateral cryptorchidism, or patients with successful spermatozoa on one side of the testis, then the oocyte retrieval and MicroTESE on the same day is most conducive to successful fertilization and pregnancy. This can eliminate the risks of freezing injury to the sperm during cryopreservation or sperm loss during the thawing procedure, thus to maximize the use of the very small amount of sperm obtained. If the rate of sperm acquisition is not high, and the spouse's chances of receiving donor sperm assisted reproduction are large, the influence of ovarian stimulation and egg retrieval on the woman and high treatment costs in IVF treatment may make the couple and doctor hesitate. At this time, it is a compromise to perform MicroTESE and freeze the sperm in advance.
Our study demonstrates that the etiology is predictive of the SRR in NOA patients. Among all the etiologic categories of NOA, NOA of definite etiology, such as cryptorchidism, has relatively high SRR. There was no significant difference in the clinical features of NOA of different etiology except KS with much smaller testicular volume. Based on our findings, using of frozen sperm from MicroTESE had similar pregnant outcome to that of fresh sperm, further illustrating the stability and feasibility of frozen sperm from MicroTESE in ICSI treatment and the value of clinical applications.
Acknowledgments
Funding: This work was supported by the National Natural Science Foundation of China (grant number 81671511 to P Ping, 81871199 and 81501310 to Y Ma, 81501243 to S Zou).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the Reproductive Ethics Committee of Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University (No. 2018072612). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Turek PJ, Givens CR, Schriock ED, et al. Testis sperm extraction and intracytoplasmic sperm injection guided by prior fine needle aspiration mapping in the nonobstructive azoospermia. Fertil Steril 1999;71:552-7. [Crossref] [PubMed]
- Hauser R, Botchan A, Amit A, et al. Multiple testicular sampling in non-obstructive azoospermia—is it necessary? Hum Reprod 1998;13:3081-5. [Crossref] [PubMed]
- Bohring C, Schroeder-Printzen I, Weidner W, et al. Serum levels of inhibin B and follicle-stimulating hormone may predict successful sperm retrieval in men with azoospermia who are undergoing testicular sperm extraction. Fertil Steril 2002;78:1195-98. [Crossref] [PubMed]
- Carpi A, Sabanegh E, Mechanick J. Controversies in the management of nonobstructive azoospermia. Fertil Steril 2009;91:963-70. [Crossref] [PubMed]
- Chen SC, Hsieh JT, Yu HJ, et al. Appropriate cut-off value for follicle-stimulating hormone in azoospermia to predict spermatogenesis. Reprod Biol Endocrinol 2010;8:108. [Crossref] [PubMed]
- Nagata Y, Fujita K, Banzai J, et al. Seminal plasma inhibin-B level is a useful predictor of the success of conventional testicular sperm extraction in patients with nonobstructive azoospermia. J Obstet Gynaecol Res 2005;31:384-8. [Crossref] [PubMed]
- Vernaeve V, Staessen C, Verheyen G, et al. Can biological or clinical parameters predict testicular sperm recovery in 47,XXY Klinefelter’s Syndrome patients? Hum Reprod 2004;19:1135-9. [Crossref] [PubMed]
- World Health Organization. WHO Laboratory manual for the examination and processing of human semen and Sperm-Cervical Mucus and interaction. Cambridge: WHO, 2010.
- Cerilli LA, Kuang W, Rogers DA. Practical approach to testicular biopsy interpretation for male infertility. Arch Pathol Lab Med 2010;134:1197-204. [PubMed]
- Dabaja AA, Schlegel PN. Microdissection testicular sperm extraction: an update. Asian J Androl 2013;15:35-9. [Crossref] [PubMed]
- Ramasamy R, Yagan N, Schlegel PN. Structural and functional changes to the testis after conventional versus microdissection testicular sperm extraction. Urology 2005;65:1190-4. [Crossref] [PubMed]
- Silber SJ. Microsurgical TESE and the distribution of spermatogenesis in non-obstructive azoospermia. Hum Reprod 2000;15:2278-84. [Crossref] [PubMed]
- Jaffe TM, Kim ED, Hoekstra TH, et al. Sperm pellet analysis: A technique to detect the presence of sperm in men considered to have azoospermia by routine semen analysis. J Urol 1998;159:1548-50. [Crossref] [PubMed]
- Vernaeve V, Tournaye H, Schiettecatte J, et al. Serum inhibin B cannot predict testicular sperm retrieval in patients with non-obstructive azoospermia. Hum Reprod 2002;17:971-6. [Crossref] [PubMed]
- Boitrelle F, Robin G, Marcelli F, et al. A predictive score for testicular sperm extraction quality and surgical ICSI outcome in non-obstructive azoospermia: a retrospective study. Hum Reprod 2011;26:3215-21. [Crossref] [PubMed]
- Plouvier P, Barbotin AL, Boitrelle F, et al. Extreme spermatogenesis failure: andrological phenotype and intracytoplasmic sperm injection outcomes. Andrology 2017;5:219-25. [Crossref] [PubMed]
- Ramasamy R, Padilla WO, Osterberg EC, et al. A comparison of models for predicting sperm retrieval before microdissection testicular sperm extraction in men with nonobstructive azoospermia. J Urol 2013;189:638-42. [Crossref] [PubMed]
- Raman JD, Schlegel PN. Testicular sperm extraction with intracytoplasmic sperm injection is successful for the treatment of nonobstructive azoospermia associated with cryptorchidism. J Urol 2003;170:1287-90. [Crossref] [PubMed]
- Haimov-Kochman R, Prus D, Farchat M, et al. Reproductive outcome of men with azoospermia due to cryptorchidism using assisted techniques. Int J Androl 2010;33:e139-43. [Crossref] [PubMed]
- Negri L, Albani E, DiRocco M, et al. Testicular sperm extraction in azoospermic men submitted to bilateral orchidopexy. Hum Reprod 2003;18:2534-39. [Crossref] [PubMed]
- Wiser A, Raviv G, Weissenberg R, et al. Does age at orchidopexy impact on the results of testicular sperm extraction? Reprod Biomed Online 2009;19:778-83. [Crossref] [PubMed]
- Ramasamy R, Ricci JA, Palermo GD, et al. Successful fertility treatment for Klinefelter’s syndrome. J Urol 2009;182:1108-13. [Crossref] [PubMed]
- Lu C, Wang Y, Zhang F, et al. DAZ duplications confer the predisposition of Y chromosome haplogroup K to non-obstructive azoospermia in Han Chinese populations. Hum Reprod 2013;28:2440-9. [Crossref] [PubMed]
- Eggers S, DeBoer KD, van den Bergen J, et al. Copy number variation associated with meiotic arrest in idiopathic male infertility. Fertil Steril 2015;103:214-9. [Crossref] [PubMed]
- Ramasamy R, Ridgeway A, Lipshultz LI, et al. Integrative DNA methylation and gene expression analysis identifies discoidin domain receptor 1 association with idiopathic nonobstructive azoospermia. Fertil Steril 2014;102:968-73.e3. [Crossref] [PubMed]
- Habermann H, Seo R, Cieslak J, et al. In vitro fertilization outcomes after intracytoplasmic sperm injection with fresh or frozen-thawed testicular spermatozoa. Fertil Steril 2000;73:955-60. [Crossref] [PubMed]
- Küpker W, Schlegel PN, Al-Hasani S, et al. Use of frozen-thawed testicular sperm for intracytoplasmic sperm injection. Fertil Steril 2000;73:453-8. [Crossref] [PubMed]
- Yu Z, Wei Z, Yang J, et al. Comparison of intracytoplasmic sperm injection outcome with fresh versus frozen-thawed testicular sperm in men with nonobstructive azoospermia: a systematic review and meta-analysis. J Assist Reprod Genet 2018;35:1247-57. [Crossref] [PubMed]
- Tavukcuoglu S, Al-Azawi T, Al-Hasani S, et al. Using Fresh and Frozen Testicular Sperm Samples in Couples Undergoing ICSI-MicroTESE Treatment. J Reprod Infertil 2013;14:79-84. [PubMed]
- Karacan M, Alwaeely F, Erkan S. Outcome of intracytoplasmic sperm injection cycles with fresh testicular spermatozoa obtained on the day of or the day before oocyte collection and with cryopreserved testicular sperm in patients with azoospermia. Fertil Steril 2013;100:975-80. [Crossref] [PubMed]


