Long-term outcomes for penile cancer patients presenting with advanced N3 disease requiring a myocutaneous flap reconstruction or primary closure—a retrospective single centre study
Introduction
Genital malignancies including squamous cell carcinoma of the penis and the scrotum are rare tumours in Europe and North America (1,2). The majority of penile cancer cases present with lesions involving the glans penis and prepuce. Therefore, the lesion is obvious to the patient and if managed early enough, penile preserving surgical techniques can be utilised with an excellent functional and cosmetic outcome (3,4). The most important predictor of survival is the presence and extent of metastatic disease in the regional lymph nodes (5-7). Provided that the inguinal lymph nodes are either impalpable or staged N1 or N2, the options available for the management of the inguinal lymph nodes include dynamic sentinel lymph node biopsy (DSNB for clinically impalpable disease), superficial modified inguinal lymphadenectomy or radical inguinal lymphadenectomy.
Rarely patients may present with advanced disease due to either an aggressive tumour subtype or more often due to a delayed presentation to the clinician. These patients present a therapeutic challenge, as inevitably they develop local infiltration of tumour into the subcutaneous tissues, skin, or the deeper structures such as the fascia lata, quadriceps muscles and femoral vessels as well as extending into the pelvic cavity.
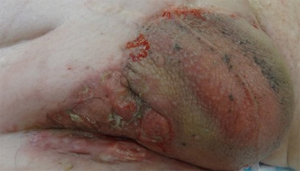
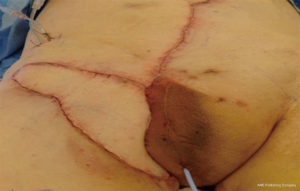
In these delayed cases the tumour mass can ulcerate through the overlying skin leading to an unsightly fungating mass or more catastrophically erosion into the adjacent femoral vessels may also occur (Figure 1). Disease extension into the lower urinary tract or rectum can also impair bowel and bladder function in these patients.
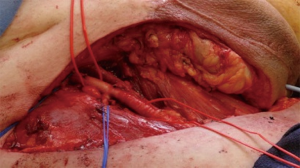
The 8th edition of the unified TNM staging for penile cancer was published in 2017 (8). This represents a consensus between representatives of the American Joint Committee on Cancer (AJCC) and the Union Internationale Contre le Cancer (UICC). In this updated edition, N3 nodal disease refers to fixed inguinal or pelvic nodes. Therefore, the presence of N3 disease commonly requires an extensive resection of the inguinal lymph nodes and overlying skin and subcutaneous tissue. The resulting defects which result from these extensive resections are often too large to allow primary skin closure and therefore reconstructive techniques are required in order to cover these defects (Figure 2). The overall prognosis for these individuals is poor, although surgical intervention is a useful palliative option which allows the patients to be discharged home with some form of urinary diversion and more manageable wounds. The surgical intervention also reduces the tumour burden and allows coverage of the areas infiltrated by the disease.
We report the surgical techniques, outcomes, complications and patient benefits in our series of patients who have presented with loco-regionally advanced penile malignancies and N3 disease.
Methods
A total of 18 patients presented to our unit with advanced genital malignancies over a 15-year period. The case records of the patients were retrospectively reviewed and data recorded onto an institutional database. The mean age of the patients was 62 (range, 37–85) years. This was a retrospective descriptive comparative study of a consecutive cohort of N3 patients undergoing standard of care management according to our institutional guidelines. This study was registered as a departmental audit.
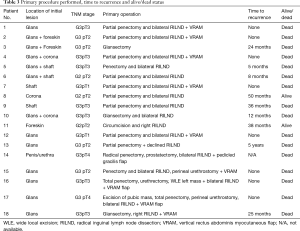
The primary tumour stage and grade for each patient is listed in Table 1.

Full table
In this series 10 patients (55.6%) presented with a primary penile cancer together with concurrent extensive nodal disease in the regional nodes who underwent tumour resection and reconstruction during the same hospital episode. The presenting symptoms (pain, discharge, poor hygiene and impaired mobility) and large tumour burden (skin and muscle involvement, fungating mass, impending erosion into femoral vessels) was felt to be too advanced to undergo neoadjuvant chemotherapy or radiotherapy based on the multidisciplinary meeting consensus. The remaining 8 patients (44.4%) presented with recurrent disease in the inguinal nodes having already undergone treatment for the primary lesion and inguinal lymph nodes. All of the patients were staged preoperatively with a staging CT scan (chest, abdomen and pelvis) to include the inguinal and pelvic lymph nodes and if deemed necessary an MRI of the penis and pelvis was performed to allow surgical planning. None of the patients within this cohort had distant metastatic disease. The tumour was assessed both pre- and intra-operatively to allow surgical planning. This included degree of skin involvement, tumour dimensions on imaging, assessing the depth and proximity to the femoral vessels in addition to any invasion into adjacent muscles and whether it was mobile or fixed.
Following resection of the primary or recurrent lesion, the resulting defect was assessed for primary closure. Defects which could not be closed primarily were covered using a combination of reconstructive techniques. Patients were subdivided into those requiring a myocutaneous flap and those who underwent primary closure. Post-operative complications were recorded according to the Clavien-Dindo classification. P value was calculated using a Chi squared test. P value of <0.05 was considered as statistically significant.
The majority of patients requiring a urinary diversion underwent a formation of a perineal urethrostomy.
Kaplan-Meier (KM) curves were calculated to analyse the cancer-specific survival (CSS) rates for this cohort of patients. KM curves were also compared with a cohort of N3 patients (n=69) who did not require flap reconstruction (Table 2). P value was calculated by log-rank test. P value of <0.05 was considered as statistically significant.

Full table
Vertical rectus abdominis myocutaneous (VRAM) flap—technique
The VRAM flap allows coverage of large defects and utilizes tissue which is well away from any previously irradiated area. The fact that it is a pedicled vascularized flap reduces the probability of flap failure provided that the inferior epigastric complex has been preserved and not involved in the disease.
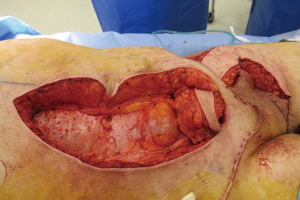
A template of the groin defect is made which is then used to mark out the size of the VRAM flap. Once marked out the skin and underlying rectus abdominis is mobilized and detached superiorly. An incision is then made along the medial border of the rectus fascia and the skin island is deepened toward the superior portion of the flap until the linea alba is seen. The contralateral rectus abdominis muscle can be visualized under the rectus fascia. The lateral incision parallels the incision in the rectus fascia keeping intact a 5-cm width of rectus fascia to be taken with the flap (Figure 3). This ensures that the perforators coming off the anterior surface of the muscle through the rectus fascia are not damaged and the underlying rectus abdominis is mobilized and detached superiorly. The inferior epigastric vessels should be seen emerging from the lateral portion of the rectus muscle. The flap is mobilized on the inferior epigastric vascular pedicle and tunneled in order to cover the groin defect (Figure 4).
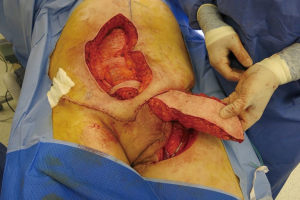
A synthetic mesh is then used to reconstruct the abdominal wall and the skin is closed primarily
Results
The case records of all of the patients in the series were reviewed.
Table 3 shows the primary procedures undertaken and the time interval to first developing recurrent disease in those patients with an interval presentation to advanced disease.
Full table
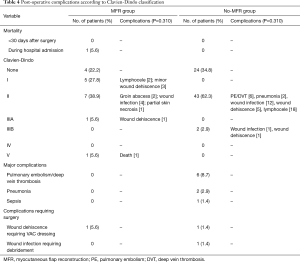
Wound related issues and surgical site infection were the main complications for this cohort and are listed in Table 4 according to the Clavien-Dindo classification. Symptomatic lymphoceles were managed conservatively with aspiration/drainage and they all eventually resolved.
Full table
The most commonly utilised myocutaneous flap involved the use of a pedicled VRAM flap (n=15). Generally, this was harvested from the contralateral side to the groin defect following resection of the inguinal mass.
A typical presentation is shown in Figure 1 which illustrates an extensive groin mass due to metastatic penile cancer. Following surgical resection, the large defect (Figure 2) is covered using a contralateral VRAM flap (Figure 5). In cases where there were smaller defects (n=3), either the tensor fascia lata (TFL) flap (unilateral or bilateral) or gracilis flap was employed. In all the cases, a pedicled flap was used.
The N3 cohort was divided into those requiring a myocutaneous flap reconstruction (MFR) and those which underwent primary closure (no-MFR).
The median length of hospital stay was 23 days (range, 10–45 days) in the MFR group. Overall, we were able to discharge all but one of the MFR patients back to their home.
Post-operatively 10 patients underwent adjuvant radiotherapy due to their extensive disease. A further 6 patients underwent adjuvant chemotherapy using cisplatin-based regimes. Five patients underwent neoadjuvant chemotherapy. Out of the 18 patients included in the MFR group, two are still alive. Two of the patients died from causes unrelated to penile cancer (cardiac arrest & pneumonia). The median survival for this cohort was 11 months.
The median length of hospital stay for the no-MFR group was 8.5 days (range, 1–38 days).
Overall, 45 patients developed various complications in the no-MFR group; ranging from pulmonary embolism, deep vein thrombosis, pneumonia to wound infection, wound dehiscence and lymphocele. Only 2 patients required a return to the operating theatre, 1 for application of a VAC dressing following wound dehiscence and the other for wound debridement due to infection.
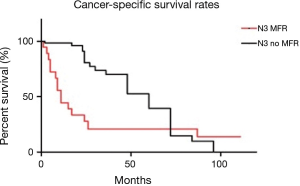
To date 25 (36.2%) patients have died from penile cancer in this group. The median survival was 60 months in the no-MFR group (Table 2). The 5-year CSS was 20.9% for the MFR group and 39.8% for the no-MFR group (P<0.01).
Figure 6 shows the disease-specific survival within the cohorts.
Discussion
Patients presenting with advanced penile cancer and extensive nodal disease have a poor prognosis and are a difficult group of patients to manage as there is very little in the way of effective chemotherapy or radiotherapy available which can achieve a complete response. In the majority of cases the surgical intervention is deemed palliative. The current EAU guidelines recommend management of patients with large and bulky inguinal lymph nodes by a multimodal approach. Neoadjuvant chemotherapy to down-size the lymph nodes followed by radical lymphadenectomy in responders is recommended although the evidence for this is limited (9). Additionally, there is a paucity of data related to the use and benefits of neoadjuvant radiotherapy regimens in cases with advanced disease. Radiotherapy for advanced lymph node disease remains a palliative treatment modality. Until the data available is more meaningful, clear guidelines are unavailable and a case by case approach is utilised for each patient. In this study, only 5 patients were deemed medically fit to undergo neoadjuvant chemotherapy due to the advanced nature of their disease. In addition, the current EAU guidelines for neoadjuvant chemotherapy in bulky fixed inguinal masses are relatively new, whereas our study spans over a 15-year period.
The aim of surgical intervention in this group of patients is to reduce the morbidity associated with an extensive ulcerating malodorous regional lymph node mass and local disease extension as well as providing a means for urinary diversion. Surgical resection of these lesions allows patients to regain a degree of mobility, allow better pain control and reduce the risk of fatal vascular invasion. In addition to this, patients have the opportunity to go home without debilitating lymphadenopathy and unsightly tumour growth and therefore have a more dignified period of survival.
Although this is a challenging group of patients to treat, we have demonstrated that in well-motivated patients with a reasonable performance status, where the disease is surgically resectable, 16/18 patients were able to be discharged home. The remaining patient was discharged to a hospice. One patient had a post-operative cardiac arrest and died within 1 week of his surgery. The reduction in the tumour burden combined with better mobility and reduced genital lymphoedema also allowed for adjuvant chemotherapy treatment to be delivered to 6 patients postoperatively.
Adjuvant chemotherapy has been utilised in a previous study involving patients with fixed inguinal lymph nodes. In all, 56% of patients with irresectable disease who underwent cisplatin and 5-FU treatment were found to be resectable (10). In patients with advanced disease the results are not so good with a 32% complete/partial response rate and 12% treatment related mortality (11). Patients presenting with fixed nodal disease as in our series were considered unsuitable for chemotherapy due to the extent of the disease and the poor performance status. One patient developed septicaemia due to the wound infection and required early surgical intervention. However, the combination of taxanes, cisplatin and 5-FU may show a better response rate in the future and thereby have a role in the neoadjuvant setting with this group of patients (9).
External beam radiotherapy is used to reduce local recurrence rates following inguinal lymphadenectomy where there is extracapsular extension. However, in cases of fixed nodal disease, radiotherapy followed by surgical debulking runs the risk of significant wound complications. The tumour burden may also be extensive, such that a large irradiated field is required. Nevertheless, in those patients with a poor performance status, radiotherapy may reduce the tumour burden and reduce symptomatic bleeding associated with large ulcerating lesions.
The most commonly performed procedure in this series involved removal of the inguinal lymph nodes/mass and coverage of the defect with a VRAM flap. This was successfully performed in 15 patients with complications being limited to lymphoceles or partial wound breakdown which healed with conservative treatment. However, in one patient the abdominal wound from the donor site broke down due to skin necrosis and highlighted the need to ensure that the flap is harvested such that the muscle and the immediate overlying skin and subcutaneous tissue is taken without extending laterally and therefore compromising the blood supply. In this particular case the wound healed by secondary intention after a prolonged period of time with the aid of vacuum-assisted closure (VAC) dressing. The VRAM flap as previously described can be mobilised sufficiently to cover large lower abdominal defects and groin defects and allows healing on a well vascularised bed (12). This is one of the largest reported series of the use of a VRAM flap to cover the inguinal wound defect following extensive inguinal tumour resection in penile cancer patients.
Alternative tissue coverage can utilise a TFL flap or gracilis pedicled flap which has been used successfully in other series to cover groin defects (13,14). With the TFL flap particularly, distal flap necrosis can be a problem especially with longer flaps and therefore we found that the larger defects were better served by using a VRAM flap.
The 5-year CSS in the no-MFR group was better (39.8% vs. 20.9%) compared to the MFR group. This is likely due to the advanced disease presentation and increased tumour burden resulting in higher risk of occult micrometastatic disease in the MFR group. Additionally, 36 (52%) patients from the no-MFR group received adjuvant chemotherapy/radiotherapy. Also, patients in the no-MFR group are likely to have a better performance status due to a lower disease burden. However, it has been reported that both chemotherapy and or radiotherapy do not improve overall survival in penile cancer patients (15). Recently, Joshi et al. demonstrated on a multivariate analysis of 1,123 penile cancer patients’ that lymph node dissection (HR, 0.64; 95% CI, 0.52–0.78; P<0.001) was associated with better overall survival, while neither chemotherapy (HR, 1.01; 95% CI, 0.80–1.26; P=0.95) nor radiotherapy (HR, 0.85; 95% CI, 0.70–1.04; P=0.11) was associated with overall survival (16). In another recent systematic review by Robinson et al., they failed to demonstrate good quality evidence to support the benefit of radiotherapy following lymphadenectomy in terms of relapse or survival (17). Ottenhof et al. studied 15 penile cancer patients with advanced disease from two centres, they demonstrated that surgical resection with reconstruction of the groin is associated with a risk of perioperative complications, including high-grade Clavien-Dindo complications. Ten of the 15 patients (67%) died of their disease, whereas 14 of the 18 patients (77.8%) in our cohort died of their disease. They concluded that surgery must be carefully considered in this group of patients as there is a lack for alternative efficacious treatment options (18).
The no-MFR group required less skin and subcutaneous tissue excision, shorter operative time, primary wound closure without tension on the skin and therefore a quicker recovery. Although, this group of patients underwent less extensive surgery with shorter anaesthetic times and hospital stay, the complication rates still remain high (Table 2). This may reflect the aggressive nature of cancers presenting with N3 disease and a poor performance status. Despite numerous advances in the last decade in the management of penile cancer, the overall prognosis of these patients remains poor (15).
Currently 11% of the patients who have undergone surgery for advanced disease are still alive. We have demonstrated that in well-motivated patients, surgical resection of highly advanced tumours is feasible and should be performed in a multimodality setting.
Our literature review found this study is one of the largest series using the VRAM flap for extensive inguinal disease in penile cancer patients from a single centre.
The limitations of our study are the retrospective nature and the relatively small cohort size. However, penile cancer is a rare cancer and cases requiring MFR are limited. Due to the retrospective design of the study, we were unable to use validated quality of life measurement tools to objectively assess any changes.
Conclusions
The options for patients with penile tumours and fixed nodal disease are limited. Current chemotherapeutic regimes show a promising yet limited response in the neo-adjuvant setting. These patients often present with a poor performance status and are unlikely to tolerate the side effects of chemotherapy. Surgical resection of these lesions with reconstruction offers both symptom control and in those patients without M1 disease, a chance for cure. With targeted therapies now increasingly being investigated, previous non-responders may now be able to reduce the tumour burden and therefore allow surgical resection. Until these options are more effective, a case by case approach is required in order to manage the cases presenting with extensive disease.
Acknowledgements
This study is supported by the NIHR Biomedical Research Centre UCLH.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Parkin DM, Muir CS. Cancer Incidence in Five Continents. Comparability and quality of data. IARC Sci Publ 1992.45-173. [PubMed]
- Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2009. CA Cancer J Clin 2009;59:225-49. [Crossref] [PubMed]
- Pietrzak P, Corbishley C, Watkin N. Organ-sparing surgery for invasive penile cancer: early follow-up data. BJU Int 2004;94:1253-7. [Crossref] [PubMed]
- Smith Y, Hadway P, Biedrzycki O, et al. Reconstructive surgery for invasive squamous carcinoma of the glans penis. Eur Urol 2007;52:1179-85. [Crossref] [PubMed]
- Ornellas AA. Management of penile cancer. J Surg Oncol 2008;97:199-200. [Crossref] [PubMed]
- Ornellas AA, Seixas AL, Marota A, et al. Surgical treatment of invasive squamous cell carcinoma of the penis: retrospective analysis of 350 cases. J Urol 1994;151:1244-9. [Crossref] [PubMed]
- Horenblas S. Lymphadenectomy for squamous cell carcinoma of the penis. Part 2: the role and technique of lymph node dissection. BJU Int 2001;88:473-83. [Crossref] [PubMed]
- Brierley JD, Gospodarwicz MK, Wittekind C. TNM classification of Malignant Tumours. 8th edition. Oxford, UK: Wiley-Blackwell, 2017.
- Hakenberg OW, Compérat EM, Minhas S, et al. EAU guidelines on penile cancer: 2014 update. Eur Urol 2015;67:142-50. [Crossref] [PubMed]
- Pizzocaro G, Nicolai N, Milani A. Taxanes in combination with cisplatin and fluorouracil for advanced penile cancer: preliminary results. Eur Urol 2009;55:546-51. [Crossref] [PubMed]
- Kattan J, Culine S, Droz JP, et al. Penile cancer chemotherapy: twelve years' experience at Institut Gustave-Roussy. Urology 1993;42:559-62. [Crossref] [PubMed]
- Kayes OJ, Durrant CA, Ralph D, et al. Vertical rectus abdominis flap reconstruction in patients with advanced penile squamous cell carcinoma. BJU Int 2007;99:37-40. [Crossref] [PubMed]
- Nirmal TJ, Gupta AK, Kumar S, et al. Tensor fascia lata flap reconstruction following groin dissection: is it worthwhile?. World J Urol 2011;29:555-9. [Crossref] [PubMed]
- Ali AT, Rueda M, Desikan S, et al. Outcomes after retroflexed gracilis muscle flap for vascular infections in the groin. J Vasc Surg 2016;64:452-7. [Crossref] [PubMed]
- Shukla CJ, Shabbir M, Feneley MR, et al. Palliation of male genital cancers. Clin Oncol (R Coll Radiol) 2010;22:747-54. [Crossref] [PubMed]
- Joshi SS, Handorf E, Strauss D, et al. Treatment Trends and Outcomes for Patients With Lymph Node-Positive Cancer of the Penis. JAMA Oncol 2018;4:643-9. [Crossref] [PubMed]
- Robinson R, Marconi L, MacPepple E, et al. Risks and Benefits of Adjuvant Radiotherapy After Inguinal Lymphadenectomy in Node-positive Penile Cancer: A Systematic Review by the European Association of Urology Penile Cancer Guidelines Panel. Eur Urol 2018;74:76-83. [Crossref] [PubMed]
- Ottenhof SR, Leone A, Djajadiningrat RS, et al. Surgical and Oncological Outcomes in Patients After Vascularised Flap Reconstruction for Locoregionally Advanced Penile Cancer. Eur Urol Focus 2018. [Epub ahead of print]. [Crossref] [PubMed]