Multispecialty retrospective review of the clinical utility of pelvic magnetic resonance imaging in the setting of pelvic pain
Introduction
The definition of male chronic pelvic pain syndrome (CPPS) is discomfort or pain in the genitalia, pelvic, or perineum region, with or without urinary symptoms, lasting at least 3 of the past 6 months (1). This encompasses a significant portion of urologic and non-urologic visits (2), with CPPS representing the primary Current Procedural Terminology code in 8% and 1% of urology and primary care visits, respectively (3). Other estimates place the prevalence at 2.5% to 16% of men younger than 50 years old (4). Such a high volume of disease places quite a financial and social burden. Current estimates place the financial burden at approximately $4,000 per patient per year (5), and the quality of life of CPPS patients has been compared to patients with congestive heart failure, Crohn’s disease, diabetes mellitus, and angina (6).
Most chronic diseases with this amount of patient distress and financial strain have a clear algorithm for diagnosis. This is not the case with CPPS. On the contrary, CPPS is a syndrome that presents to numerous specialties, including neurology, orthopedics, gastroenterology, pain medicine, and urology, each with a different treatment algorithm (7-11).
Knowing that the workup for male CPPS lacks guidance across specialties, we were curious about imaging practices and diagnostic utility. Upon reviewing the literature, there is a paucity of data in the imaging workup of chronic pelvic pain. There are a few studies of women with chromic pelvic pain, in which pain appears to be adequately evaluated with MRI (11,12). However, there is no data outlining the role of computed tomography (CT) scan, transrectal/abdominal ultrasound, or pelvic MRI into assess male chronic pelvic pain. The aim of our study was to evaluate the use of pelvic MRI in men whose chief complaint is chronic pelvic pain.
Methods
After receiving IRB approval, we performed a retrospective review of all pelvic MRIs performed on men at our institution from January 2, 2010, to December 31, 2014. We delineated the MRIs to studies ordered using pelvic pain-related International Classification of Diseases, Ninth Revision codes (penile pain, testicular pain, scrotal pain, male pelvic pain, prostatitis, hip pain, rectal pain, joint pain, and orchitis). In further chart review, the radiology final impression was analyzed to determine whether anatomic abnormalities were present on MRI. We also determined the specialty of the ordering provider for each exam, and these were grouped into urology, medicine subspecialties, gastroenterology/colorectal surgery, orthopedics/neurology, pain management, and oncology. Each of these specialties was analyzed for both positivity on MRI and pre-existing diagnosis of oncologic disease.
Results
During the study period, 2,643 non-musculoskeletal male pelvis MRIs were performed at our institution. Of these, 597 (23%) were ordered for male pelvic pain. Results by specialty are listed in the Table 1. Overall congenital or acquired abnormalities were identified in 205 of 597 cases (34%). The most commonly recognized abnormalities were prostatitis, bony fractures, osteomyelitis, labral tears, proctitis, perianal fistulas, inguinal hernias, or oncologic lesions. Only 5% of MRIs for male pelvic pain were ordered in the setting of malignancy; not surprisingly the majority of those patients were followed by oncologists. In our database, the vast majority of MRIs were ordered by pain management services, and in general, yield of MRI declined as the number of total tests increased.
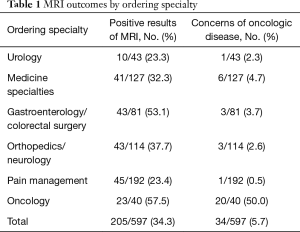
Full table
Discussion
With such a wide array of specialties involved, developing a consensus algorithm to guide the diagnosis of CPPS is difficult. Most diagnosis/treatment is based on physical exam, questionnaire, microbiologic testing, and trials of drug administration (8), often not revealing the etiology of the pain. Very little data are available to suggest the imaging modality of choice in chronic pelvic pain. The obstetrics gynecology and female chronic pelvic pain literature has data to suggest that MRI is helpful in finding endometriosis (a common cause of female chronic pelvic pain), but no such studies have been reciprocated for men (11,12).
The clinical utility of imaging in male pelvic pain can be thought of in 3 categories. Category 1 is to diagnose the causation of the pain. Our data showed that MRI had a positive result in 34% of all subsets of patients. Taken into context with other screening modalities, this is actually quite an impressive number. Microscopic hematuria has an accepted imaging finding of 5% on CT (13). We are aware that this is not a perfect comparison secondary to the primary aim of detecting malignancy in the hematuria screening population versus the unlikelihood of cancer in the pelvic pain population. This comparison is further distorted when considering the increased costs associated with MRI testing when compared with CT scan. Other chronic pain syndromes, such as chronic back pain, are also difficult to compare. The frequency of incidental findings on MRI in the chronic back pain population has been reported as high as 52% in patients (14). With similar regard, we acknowledge that a limitation of our study is that we are assuming that imaging findings are the likely causation of patient pain. Since this study does not correlate clinical treatment with improvement of pain subscores, we are unable to assess the level of incidental findings in our patient subset.
The second category of clinical significance in the MRI patient population is the ability to reassure the patient. MRI is a very specific test, and by obtaining a negative result in 66% of patients, you are able to provide reassurance to the patient that there is no anatomic or oncologic pathology. Though not diagnostic, it provides clarification in the work-up of pelvic pain and provides the patient with reassurances against a catastrophic diagnosis.
The final category involves the algorithm in which MRI fits into the CPPS treatment paradigm. Given the cost related to MRI and the unknown significance of incidental findings, it is our suggestion that MRI be reserved for refractory cases of CPPS. This will limit the addition of an expensive modality to an already cost prohibitive disease pathology. This can be further enhanced by future studies to determine the usefulness of more economic imaging modalities such as CT and ultrasound.
Conclusions
CPPS is a complex diagnosis with little data dictating the ordering of imaging. MRI appears to be an acceptable imaging modality in the setting of refractory pelvic pain cases. We would not recommend utilizing pelvic MRI as a screening modality. Further study needs to be undertaken to define the role of all imaging modalities in CPPS, but in the interim, pelvic MRI may provide answers in recalcitrant patients.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: After receiving IRB approval (IRB 15-004216), we performed a retrospective review of all pelvic MRIs performed on men at our institution from January 2, 2010, to December 31, 2014.
References
- Pontari MA, Ruggieri MR. Mechanisms in prostatitis/chronic pelvic pain syndrome. J Urol 2004;172:839-45. [Crossref] [PubMed]
- Nickel JC, Downey J, Hunter D, et al. Prevalence of prostatitis-like symptoms in a population based study using the National Institutes of Health chronic prostatitis symptom index. J Urol 2001;165:842-5. [Crossref] [PubMed]
- Murphy SF, Schaeffer AJ, Done J, et al. IL17 Mediates pelvic pain in experimental autoimmune prostatitis (EAP). PLoS One 2015;10:e0125623. [Crossref] [PubMed]
- Falahatkar S, Shahab E, Gholamjani Moghaddam K, et al. Transurethral intraprostatic injection of botulinum neurotoxin type A for the treatment of chronic prostatitis/chronic pelvic pain syndrome: results of a prospective pilot double-blind and randomized placebo-controlled study. BJU Int 2015;116:641-9. [Crossref] [PubMed]
- Duloy AM, Calhoun EA, Clemens JQ. Economic impact of chronic prostatitis. Curr Urol Rep 2007;8:336-9. [Crossref] [PubMed]
- Wenninger K, Heiman JR, Rothman I, et al. Sickness impact of chronic nonbacterial prostatitis and its correlates. J Urol 1996;155:965-8. [Crossref] [PubMed]
- Antolak SJ Jr, Hough DM, Pawlina W, et al. Anatomical basis of chronic pelvic pain syndrome: the ischial spine and pudendal nerve entrapment. Med Hypotheses 2002;59:349-53. [Crossref] [PubMed]
- Rees J, Abrahams M, Doble A, et al. Diagnosis and treatment of chronic bacterial prostatitis and chronic prostatitis/chronic pelvic pain syndrome: a consensus guideline. BJU Int 2015;116:509-25. [Crossref] [PubMed]
- Vermeulen W, De Man JG, Pelckmans PA, et al. Neuroanatomy of lower gastrointestinal pain disorders. World J Gastroenterol 2014;20:1005-20. [Crossref] [PubMed]
- Woodworth D, Mayer E, Leu K, et al. Unique microstructural changes in the brain associated with urological chronic pelvic pain syndrome (UCPPS) revealed by diffusion tensor mri, super-resolution track density imaging, and statistical parameter mapping: a MAPP network neuroimaging study. PLoS One 2015;10:e0140250. [Crossref] [PubMed]
- Juhan V. Chronic pelvic pain: An imaging approach. Diagn Interv Imaging 2015;96:997-1007. [Crossref] [PubMed]
- Bharwani N, Tirlapur SA, Balogun M, et al. MRI reporting standard for chronic pelvic pain: consensus development. Br J Radiol 2016;89:20140615. [Crossref] [PubMed]
- Shinagare AB, Silverman SG, Gershanik EF, et al. Evaluating hematuria: impact of guideline adherence on urologic cancer diagnosis. Am J Med 2014;127:625-32. [Crossref] [PubMed]
- Jensen MC, Brant-Zawadzki MN, Obuchowski N, et al. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med 1994;331:69-73. [Crossref] [PubMed]


