The penoscrotal surgical approach for inflatable penile prosthesis placement
Introduction
Penile prosthesis implantation is one of the few operations in urology that has not dramatically changed over the last 44 years since it was invented in 1973 by Scott et al. (1). Although several implantation approaches have been described, the penoscrotal approach is by far the most common accounting for >80% of inflatable penile prostheses (IPPs) placed worldwide (2). Dr. Köhler’s technique described herein is a descendant of the teachings and methods of Dr. Steven K. Wilson. Hence, this method has evolved from the experience of about 12,000 penile implants—whereby ample experiences (both good and unpleasant) have caused it to evolve. In comparison to the infrapubic approach, we feel our approach offers excellent corporeal exposure in all patients, even the obese. In addition, the penoscrotal incision affords proximal crural exposure, if necessary, prevents pump migration, does not leave a scar and avoids possible neurovascular bundle injury. Most importantly, artificial urinary sphincters (AUS) can be done through the same incision and exposure (3). This is a tremendous advantage in patients who require multiple AUS revisions. The disadvantages of this approach are blind placement of the reservoir and increased scrotal swelling when compared to the infrapubic incision. Recommendations offered in the paper reflect opinions of Dr. Gupta and Dr. Köhler and may not reflect the nuanced technique of the other co-authors.
Patient selection
A surgery performed with perfect surgical technique can still disappoint a patient with the wrong or unrealistic expectations. The first article of this special edition elegantly details appropriate patient selection and counseling—we highly recommend you read this before booking patients for surgery. Distilling our practices down to a few principles would include heeding the advice of Dr. Wilson: “never implant a stranger”, Dr. Köhler: “Under-promise, and over deliver” and Dr. Trost: “Beware the CURSED Patient” (Compulsive, Unrealistic, Revision, Surgeon shopping, Entitled, Denial, Psychiatric Disorders) (4).
Documentation prior to surgery must include: failure or contraindication of phosphodiesterase-5 inhibitors, discussion of vacuum devices and injections and inquiry about Peyronie’s disease. Discussion and medical record documentation concerning possible loss of penile length, infection rates, mechanical reliability, and injury to adjacent structures (urethra, bladder, bowel, vessels) is also mandatory. There are only three true contraindications listed on the package insert of penile implants: (I) active infection present anywhere in the body, especially urinary tract or genital skin infection; (II) patients unwilling to undergo any further surgery for device revision; and (III) unresolved problems affecting urination (such as an elevated residual urine volume secondary to bladder outlet obstruction or neurogenic bladder).
Surgical technique
Positioning, prep, draping
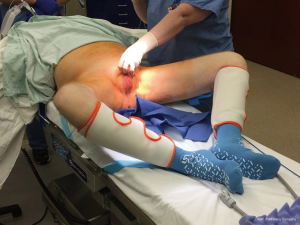
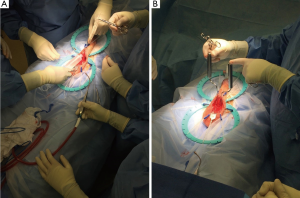
Antibiotic infusion is started in the holding area, approximately 1 hour before the operation. Once anesthesia is induced with the patient supine on the operating table, the patient’s legs are placed in the frog-leg position. Clippers are used to clip the hair on the penis, scrotum, and suprapubic area, taking care to avoid cuts. An alcohol-based prep is then used for skin prep from the mid-thighs inferiorly to the umbilicus superiorly, including the penis and scrotum. The scrotum should be held up and off the underlying skin to allow the prep to dry to achieve maximal sterilization (Figure 1).
Draping is then done while exposing the penis, scrotum, and lower abdomen. We use waterproof paper drapes rather than the ubiquitous blue cloth towels because towels tend to get wet allowing perineal organisms to possibly contaminate the operative field. Theoretically, they also drop lint on the field. After straightening the patient’s legs, a laparotomy drape with a small opening is centered on the penis and scrotum. A clear 3M® U-drape isolates the surgical field from surrounding skin and acts as a waterproof barrier and catches any fluid or irrigation that is used (Figure 2).
Trendelenburg position is used to place the penis and scrotum in a horizontal plane with the abdomen sloping downward, which creates tension on the abdominal musculature.
Retractor set-up and exposure
Proper exposure and retractor set-up are extremely important. A good set up makes the case very straightforward. Poor exposure obscures anatomy, making the case longer and more difficult and increases the chances of inadvertent injury or infection.
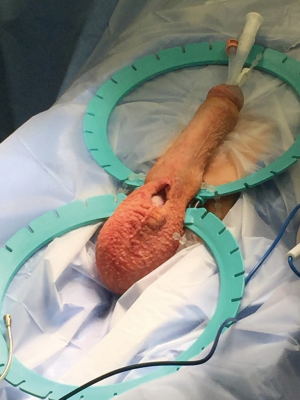
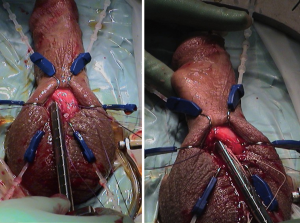
The patient is catheterized after draping is complete and the indwelling catheter is used to empty the bladder and to designate the position of the urethra by palpation during the surgery. An empty bladder is crucial for standard, retropubic placement of the reservoir to minimize the possibility of bladder injury. The Foley catheter is plugged with a catheter plug once the bladder is empty. Dr. Scott’s metal, self-retaining retractor has been revised by Dr. Wilson in disposable form and is available from both implant providers: the “SKW” deep scrotal retractor from Boston Scientific and the “Wilson” scrotal retractor from Coloplast. Each company also provides all the hooks, the strap and the baby Deavers with the retractor. The retractor is placed with the hinges at the penoscrotal junction. The beaded strap, used to present the corpora cavernosa forward, is placed in the first groove of the upper ring with 2 beads inside the ring on either side of the bar. The sharp hook is used to place the penis on traction by placing the hook inside the meatus and securing the penis toward the 12 o’clock position, creating traction on the penis over the beaded strap (Figure 3). This traction stretches the corpora and brings the proximal corpora cavernosa into the field.
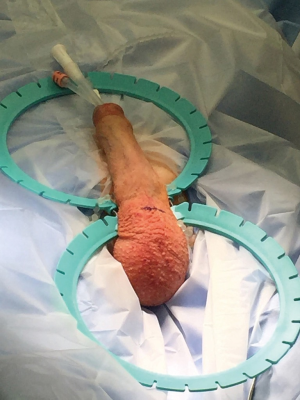
An incision is made transversely about 1–2 cm below the penoscrotal junction to a total length of about 4-cm. The incisional width can be minimized as it usually stretches when the exposure hooks are applied. When planning to perform a scrotoplasty during closure, keep in mind that the width of the incision and the height in relation to the penoscrotal junction determine the amount of tension on the scrotoplasty. We prefer a lower incision for scrotoplasty to minimize tension on the closure after scrotoplasty.
The incision is carried deep with electrocautery through the dartos. The proper depth is encountered when a hole appears through the dartos. This allows only the aperture of the incision to be brought onto the penoscrotal junction, leaving behind unnecessary layers (Figure 4).
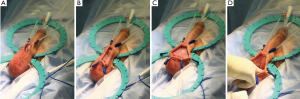
The incision is then held in place over the base of the penis and retracted open with blunt hooks. First 2 hooks are placed superiorly at 11 and 1 o’clock with loose tension, then 2 hooks are placed inferiorly at 5 and 7 o’clock with medium tension, then 2 hooks are placed laterally at 3 and 9 o’clock with tight tension (Figure 5A-C).
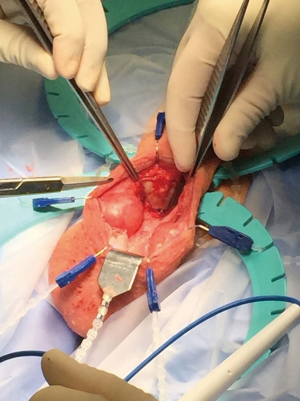
Using the index and middle fingers, with blunt dissection the scrotal contents are dissected away, revealing the proximal corpora cavernosa and urethra [the “cobra kai maneuver according to Dr. Köhler; the “Henry Sweep” according to Dr. Henry (5)] (Figure 5D). The overlying dartos tissue overlying the corpora cavernosa is then sharply dissected, revealing the “squeaky clean”, gleaming white tunica albuginea. The white tunica albuginea should be clearly visible with well-defined lateral edges of the corporal bodies (Figure 6).
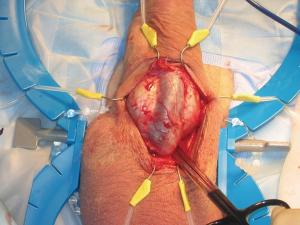
In order to achieve ample exposure of the proximal corpora cavernosa, we recommend separating the scrotal septum from the urethral attachment. This maneuver allows access to the proximal corpora in case of difficulty with placement of the cylinder bases. This maneuver also facilitates placement of the pump and allows access to the proximal urethra for simultaneous AUS placement through the same scrotal incision (3). One or more rake hooks retract the scrotum inferiorly with a rolled sponge (per Dr. Wilson, the “cowboy blanket roll”). The sponge spreads the tension of the Rakes, cushioning the scrotal contents (Figure 7).
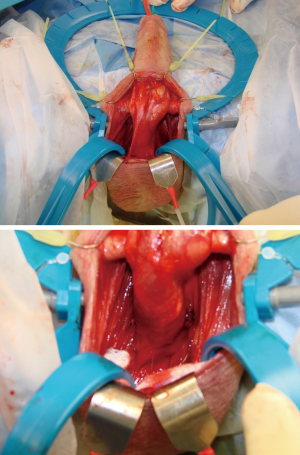
Corporotomy
The corporotomies should be proximal enough on the corpus cavernosum to ensure the tubing exits the corporotomy into the scrotum, not the penile skin. Protect the urethra while placing the stay stitches and making the incision (Figure 8).
The 2-0 vicryl stay sutures are placed around the intended corporotomy on each corpus cavernosum. The superior portion of the stitch should be below or even with the beaded strap, preventing tubing visible in the penis while maintaining ease of distal dilation. The 1.5-cm corporotomies are made with a 15-blade on each side, between the stay stitches. For a Coloplast device, corporotomy should just accept the 12 Fr Brooks dilator. For a Boston Scientific device, the corporotomy will have to be slightly larger. If the corporotomy is too small, extend the incision inferiorly, not towards the distal penis.
Distal dilation is begun with scissors or the suction tip to about 50–75% of the way to the glans penis. The instrument should deform the lateral wall of the penis to prevent crossover (Figure 9).
A 12 Brooks dilator completes the dilations proximally and distally on each side. Distally we ensure the dilator points slightly laterally to avoid crossover throughout the dilation. Squeezing the glans prevents distal perforation.
The proximal dilation is done following the path of the crura of the corpora as they flare laterally onto the pubic bone. Thus, when dilating, the dilator should trend laterally. In order to check for urethral injury after the distal dilation, forceful injection of fluid with a bulb syringe is done on each side distally while observing for urethral discharge (Figure 10A). The “field-goal test” checks the proximal dilation by placing a dilator on each side proximally. The dilator handles should be parallel and of equal depth penetration (Figure 10B). The measuring tool is used to measure the proximal and distal corpora to the most proximal stay suture. Device is then prepared on a back table. Routinely our trained operative staff prep the device in combination with the device representative’s observation. We feel it is very important that the operating surgeon should also be knowledgeable and skilled in prepping the device.
Reservoir placement
Standard placement in the retropubic space
An S retractor or baby Deaver is placed in the external inguinal ring retracts the anterior wall of the inguinal canal anteriorly, placing tension on the abdominal layers below. In order to reliably find the inguinal ring, the pubic tubercle is cleaned off and the index finger is pushed down and over the bone toward the ipsilateral shoulder to drop into the ring. With an index finger, not an instrument, the abdominal wall layers are bluntly dissected. Consistent anterior retraction and frequent repositioning of the retractor through each layer create abdominal wall tension and cause an easier dissection. Anterior retraction pulls the fascia up, making the fascia susceptible to puncture bluntly. For tough, strong fascia the finger can be twisted in a drill motion to find a weak spot where fibers give way. When through, the finger can feel the posterior side of the pubic bone. The reservoir then goes into the space that was created, leaving the rigid plastic head at the mouth of the opening into the retropubic space. To, hopefully, avoid lacerating a pelvic vessel, we only use a finger to perforate the fascia. While we strongly believe in the finger for safety, most large volume implanters use an instrument such as scissors, tonsil clamp, or nasal speculum to perforate the transversalis fascia allowing access to the retroperitoneal space. A finger is surprisingly effective with proper retraction. In our hands, if a finger cannot pierce the fascia, then the reservoir is placed ectopically.
Ectopic (aka intraabdominal wall or high submuscular)
Our preferred reservoir is the Coloplast 125 cc cloverleaf reservoir which we intentionally under-fill (to minimum cylinder requirements) to prevent an obvious bulge. The conceal reservoir by AMS can also be used but needs to be handled carefully with protected clamps. All overlying tissue is cleaned off of the pubic tubercle with a finger or Metzenbaum scissors. The tubercle must be free of all overlying Scarpa’s fascia to avoid reservoir placement superficial to the musculature. An S retractor or baby Deaver is then placed in the external inguinal ring above the cleaned tubercle, retracting the inguinal canal anteriorly. This maneuver differs from space of Retzius placement outlined above. In standard reservoir placement, the ring is pulled cephalad, resulting in a taut “drum head” outlined by the bony pelvis. In ectopic placement, the ring is lifted to the sky to for entry into the abdominal wall structures.
After entering the space above the transversalis fascia, adequate space in a superior location is created with a long grasping forceps (ring forceps, lung grasping forceps, long nasal speculum or Wilson ectopic reservoir clamp). The clamp then grasps the distal-most aspect of the reservoir and maneuvers the reservoir as superiorly as possible within the space that was created (Figure 11). It is extremely important that the pubic tubercle must be “squeaky clean” of all overlying tissue, i.e., Scarpa’s fascia, when placing an ectopic reservoir. Otherwise the reservoir will be placed in a subcutaneous location instead of submuscular, creating a visible and palpable bulge in the abdominal wall.
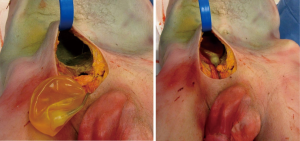
Cylinder sizing and placement
Rules of thumb for cylinder selection
Coloplast: “The rule of 10”
Proximal length minus 10 = length of rear tip extenders necessary to provide adequate tubing length outside of corporotomy. Device size is the remaining total length after subtracting the necessary RTE from the total length of cylinder necessary.
Boston Scientific
Total length minus 2 = maximum device size without rear tips. If proximal measurement is at least 3-cm longer than the distal measurement, drop down by 1 cylinder length.
A good tip when using Boston Scientific for implantation through a penoscrotal incision: rather than downsizing the Boston Scientific implant, infra-pubic sets can be used for peno-scrotal cases. The infra-pubic sets have longer tubing length from the cylinders, allowing for better pump positioning in the scrotum. However, this requires tailoring the excess length and using 2 additional quick connects to connect the cylinders to the pump.
After preparation, the implant is properly positioned on the operating table with the release button on the pump anterior and the exit tubing taking off anteriorly from the cylinders. A Keith needle and the Furlow insertion tool puncture the guide strings for each cylinder through each distal corpus cavernosum and the glans. The strings are tagged with a Kelly clamp after puncture. Both guide strings should be in position before either cylinder is put into place. A cylinder placed before the contralateral guide string is passed through the glans risks inadvertent perforation of the previously placed cylinder. After both guide strings are pierced thorough the glans, the cylinders are placed one at a time into each corpus cavernosum. The proximal end of the cylinder with RTE is first placed proximally through the corporotomy. The guide string then slides the distal end into place. Using the back of a DeBakey forceps “like a shoe horn” can be help manipulate the proximal end into place in the proximal crus. After each cylinder is placed the stay sutures are used to close the corporotomy in horizontal mattress fashion. Do not spend extra time perfectly laying out each cylinder within its corpus cavernosum. Similarly, if there is a knuckle of cylinder visible at the corporotomy indicating distal or proximal seating was insufficient, ignore it. Inflation will position the cylinders properly within the cavernosal spaces.
Tubing connections and device testing
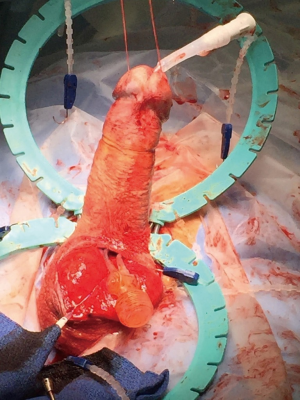
Once the corporotomies are closed the pump is connected to the reservoir. Often the perceived tubing length from cylinders to pump will decrease after inflation as the proximal cylinders are pulled down when seating. The connectors are used to connect the two ends of the reservoir tubing. The device is then inflated to seat the cylinders correctly and to test the device. The guide strings help position the distal ends of the cylinders underneath the glans if pulled during the initial inflation (Figure 12). When satisfied with the position, we keep the device inflated to about 60% for 24 hours to aid in hemostasis.
Pump placement and closed suction drainage
We place a 10-French Blake drain through the developed pump space prior to placing the implant to avoid injury to the device upon drain placement. While some surgeons have the drain exit via the pubic skin to avoid an exit wound adjacent to the pump, we prefer the exit of the drain remain dependent as this allows for discharge of additional fluid after the drain is removed should such fluid continue to collect. We then place the pump in this pocket contiguous to the drain and “lock” the pump with vicryl. With the pump in the desired location, the non-dominant hand pushes forward the posterior scrotal wall on either side of the tubing neck is it exits the pump. A single suture of 00 vicryl approximating the posterior scrotal wall tissue over the pump tubing keeps the pump and its tubing buried.
Closure
A 2-0 vicryl is used to close the deep dartos layer with a gathering stitch, with careful attention is to keep the device tubing in the scrotum and preventing its migration under the skin of the penis. Often it is useful to allow tubing to peak through the center of the gathering stitch, decreasing tension on the tubing within the scrotum and preventing tubing migration onto the penile shaft. A 3-0 monocryl stitch is used to close a more superficial dartos layer. A 4-0 monocryl then is used to close skin in simple, interrupted fashion. Dermabond is placed on the incision. Closure that breaks down from excessive tension or lack of multiple layers results in device exposure and infection. It is important to take care during closure with good technique. A “mummy wrap” is created around the penis and scrotum with a large Kerlix (5) along with fluffs and a supportive undergarment. We leave the Foley catheter in place for 24 hours.
Post-operative care
We typically leave the device about 60% inflated after implantation. This in combination with the drain and mummy wrap minimizes hematoma formation. We remove the catheter and the drain after 24 hours as long as the drain puts out less than 50 cc over 8 hours. The drain is not sewn in and the catheter is accessible through the dressing (a hole cut into the supportive undergarment) which facilitates removal of these items without disturbing the dressing as the patient removes 48 hours after surgery. Use of liposomal bupivacaine during the case aids in pain post procedure and minimizes narcotic use (6). We give 10 days of post-operative Bactrim although this practice admittedly has no evidence and probably treats the surgeon more than the patient. We write for 40 oxycodone tablets and encourage the patient to err on the side of taking them and moving about rather than lying motionless in pain. The first post-operative visit at 3–6 weeks (depending on the patient’s distance from our office) entails device cycling by the physician and implant operation teaching for the patient. About half of our patients are using the device at 1 month for sex, the other half at 2 months. We strongly recommend the patient inflates the device maximally daily for 10 minutes in the first year after surgery hoping to improve penile length (7).
Complications and lessons learned
We reviewed Dr. Köhler’s first 240 cases for which we have at least 30-month follow-up. Of the 240 cases, 140 cases were Coloplast and 100 were AMS. This is an academic practice where different level urology residents (typically 3rd years) and fellows actively participated. Case volume is mixed but about 80% of cases were primary implants. We believe our data and patient follow up information to be fairly accurate as no high-volume implanters are in the immediate vicinity (patients may go elsewhere if they are dissatisfied). However, we did not routinely telephone our patients to check their status so these complications are likely an underestimate—like most listed in the literature with attrition bias.
Five-year survival from mechanical failure: 95.4%
This was our most common complication. Device malfunction occurred in 11 patients within the follow up period: 9 AMS vs. 2 Coloplast (P=0.0092). Thus, the often quoted 95% reliability at 5 years was true in our experience. The main difference in reliability occurred from pump issues: five AMS pumps became “frozen” and unusable (Figure 13). Intraoperatively, once the scrotum was opened and the pump freed from its capsule, the device worked well without need for further revision. Of the 5 pump revisions, 2 were replaced with new AMS pumps of which one had continued intermittent pump problems. The other three patients were switched to a Coloplast pump where we wash out the space and leave the rest of the AMS device in situ. One of the pump revisions later returned with a fungal infection and underwent successful malleable substitution. Two Conceal® reservoirs developed leaks, 1 AMS cylinder developed a leak, and 3 (2 Coloplast Titan/1 AMS LGX) patients developed cylinder aneurysm. All the aneurysms occurred in revision cases where there was weakened tunica of which we were unaware. Because of the mechanical failures, we no longer use the Conceal® reservoir in our practice and prefer AMS CX cylinders in men with known corporal weakness.
Five-year survival from infection: 96.7%
Eight patients presented with infection for a 3.3% overall rate. All cases utilized infection retardant components. Seven of the eight patients were diabetic. In his entire career, Dr. Köhler has seen only one non-diabetic infection. Three of the infections were fungal and presented as “wimpy” infections with pump fixations and insidious onset that occurred >6 weeks after the surgery. The other 5 infections were “aggressive” within 6 weeks of surgery. In seven of the eight patients, malleable salvage was attempted after a two-day hospitalization for intravenous antibiotic administration. We believe this delayed salvage is necessary to allow the IV antibiotics to convert the systemically infected patient to an infection only confined to the implant spaces. Nevertheless, three of the seven malleables became re-infected and were removed. Our malleable success rate is much lower than previous reported literature (8). This is likely due to attempting salvage in “aggressive” infection with “fire engine” red erythema, pus, elevated WBC and systemic symptoms (fevers/chills/night sweats). One of the salvage procedures failed because the salvage was performed “emergently” and was not given 48 hours of IV antibiotics. We believe in the era of the coated implant, there is no such thing as an emergent salvage—either the device should be removed because the patient is sick, or admission with 2–3 days of observation of infection progression should guide treatment. We have tempered our enthusiasm of offering salvage to virulent infections. Our practice mimics others in this regard. A recent study of implant infections in the USA found only 15% were offered salvage (9).
Urethral injuries: 1.7%
We have sustained 4 urethral injuries. One was a result, as Dr. Wilson is fond of saying, a “QBM” (quick birdlike movement) during dilation in a fibrotic corpora revision case. The other 3 were sustained from penile modeling for Peyronie’s disease. One was recognized intraoperatively as the cylinder exploded out of the urethra. This case was aborted (leaving no implant material implanted) and reattempted 3 months later with apparent success. Interestingly, this patient then presented 1 year later requiring distal revision of the opposite side for impending glans erosion. Our second urethral injury was noted POD 1 with the device extruding out the urethra. The offending cylinder was removed that day and the patient presented 3 weeks later with the other cylinder coming out of the urethra. The rest of the device was removed but the patient complained of intermittent night sweats for months afterward. After an extensive work-up ruling out malignancy, the patient’s saga finally ended 6 months later after presenting with a formidable prostate abscess that required transurethral resection. Our final injury was also noted POD 1 with the cylinder coming out of the urethra. In this case we modeled against a cylinder that was appropriate for the non-shortened side but 1 cm longer than the shorter sized. Outside of our series we have encountered several delayed distal urethral injuries. Careful review of these patients’ charts revealed penile modeling as long as 3 years prior. Because of these urethral injuries, we no longer utilize modeling as an adjunctive procedure for penile straightening instead opting for pre-placed plication sutures or grafting with Tachosil® for significant Peyronie’s disease. We also never over-size cylinders in patients with Peyronie’s and use the same size cylinders on both sides despite the disparity of measurement commonly seen in this disease.
Bladder injuries: 0.8%
Our first bladder injury occurred trying to reuse a reservoir that was part of an implant which failed due to fluid leak at the pump tubing. The reservoir was empty for only a week as the patient presented with a malfunctioning device promptly. Regardless, when we inflated the reservoir, the patient bucked after 15 cc. Vigorous bladder irrigation was clear. We did not perform cystoscopy. At the end of case the patient’s urine turned bright red. We repaired the bladder and placed a new reservoir on the other side. The patient did well. Our second injury occurred after placing a reservoir in the space of Retzius following robotic prostatectomy. We counselled against this for fear of bowel complications but the patient was very worried about an unsightly bulge from an ectopic reservoir. He complained of glass shard like pain in the area of the reservoir post-operatively. Six weeks the later the patient presented with gross hematuria. This bladder injury was repaired through a midline incision and a new reservoir placed ectopically. He did well thereafter. We no longer re-use reservoirs. We follow the practice deemed by Morey as “Drain and Retain” leaving the old reservoir in place and placing a completely new system with its reservoir in a different location (10). We also do not give the patient a preference on ectopic versus traditional placement after robotic prostatectomy.
Miscellaneous complications
Proximal perforation was noted in 6 cases (2.5%). All of these were treated at the time of injury with a suture sling as described by Wilson without incident of further complication (11). Two patients (1.7%) required revision for pump repositioning as the pump had migrated into the perineum and was difficult to manipulate. One patient was returned to the OR for correction of SST deformity (0.004%). One device was removed for chronic pain (0.004%). Urinary retention occurred in about 4% of men, with most resolving with alpha blockers and a few additional days with a catheter. Since adapting our pump locking stitch, we have not had any problems with pump position. The need for true SST deformity correction is extremely rare. In our experience, most SST deformities are a result of undersized cylinders. We have also noted that uncircumcised men more commonly have an SST like appearance.
Overall surgical revision rate: 11.3%
Thus, 27 patients required a second operation within the follow-up period of our study. Mechanical problems were the most commonly encountered complication. We are very fortunate not to have experienced a vascular or bowel injury yet as most high-volume implanters are visited by one of these nightmares. We feel this likely stems from perforating the transversalis fascia with a finger only, and not hesitating to place the reservoir ectopically when difficulty is encountered or when the patient has a history of abdominal surgery such as robotic prostatectomy or cystectomy.
Conclusions
Our technique continues to evolve as complications and unpleasant outcomes are a harsh teacher. We include our detailed results and mistakes from an academic practice to hopefully help future surgeons avoid similar mishaps. Independent of complications, excellent patient selection and detailed pre-operative counseling are the best defense against unhappy patients and ensure our own personal job satisfaction.
Acknowledgements
None.
Footnote
Conflicts of Interest: Dr. Wilson is a consultant for both American Medical Systems/Boston Scientific and Coloplast; Dr. Köhler is a consultant for both American Medical Systems/Boston Scientific and Coloplast. Other authors have no conflicts of interest to declare.
References
- Scott FB, Bradley WE, Timm GW. Management of erectile impotence. Use of implantable inflatable prosthesis. Urology 1973;2:80-2. [Crossref] [PubMed]
- Trost LW, Boonjindasup AG, Hellstrom WJ. Comparison of infrapubic versus transcrotal approaches for inflatable penile prosthesis placement: a multi-institution report. Int J Impot Res 2015;27:86-9. [Crossref] [PubMed]
- Wilson S, Delk J 2nd, Henry GD, et al. New surgical technique for sphincter urinary control system using upper transverse scrotal incision. J Urol 2003;169:261-4. [Crossref] [PubMed]
- Trost LW, Baum N, Hellstrom WJ. Managing the difficult penile prosthesis patient. J Sex Med 2013;10:893-906. [Crossref] [PubMed]
- Henry GD. The Henry mummy wrap and the Henry finger sweep surgical techniques. J Sex Med 2009;6:619-22. [Crossref] [PubMed]
- Cotta BH, Welliver C, Brahmamdam A, et al. Long-acting liposomal bupivacaine decreases inpatient narcotic requirements in men undergoing penile prosthesis implantation. Turk J Urol 2016;42:230-4. [Crossref] [PubMed]
- Henry GD, Carrion R, Jennermann C, et al. Prospective evaluation of postoperative penile rehabilitation: penile length/girth maintenance 1 year following Coloplast Titan inflatable penile prosthesis. J Sex Med 2015;12:1298-304. [Crossref] [PubMed]
- Gross MS, Phillips EA, Balen A, et al. The Malleable Implant Salvage Technique: Infection Outcomes after Mulcahy Salvage Procedure and Replacement of Infected Inflatable Penile Prosthesis with Malleable Prosthesis. J Urol 2016;195:694-7. [Crossref] [PubMed]
- Zargaroff S, Sharma V, Berhanu D, et al. National trends in the treatment of penile prosthesis infections by explantation alone vs. immediate salvage and reimplantation. J Sex Med 2014;11:1078-85. [Crossref] [PubMed]
- Cefalu CA, Deng X, Zhao LC, et al. Safety of the "drain and retain" option for defunctionalized urologic prosthetic balloons and reservoirs during artificial urinary sphincter and inflatable penile prosthesis revision surgery: 5-year experience. Urology 2013;82:1436-9. [Crossref] [PubMed]
- Wilson SK. Rear tip extender sling: a quick and easy repair for crural perforation. J Sex Med 2010;7:1052-5. [Crossref] [PubMed]