Vasectomy reversal for post-vasectomy pain syndrome
Introduction
Vasectomy is a safe and effective means of providing long-term contraception to those men that choose to undergo the procedure. While techniques vary, the procedure typically involves isolation and exposure of both vasa, division of the vasa, and a method to prevent recanalization. Methods to ensure occlusion can include the use of clips, fascial interposition, ligation or mucosal cautery (1). Urologists in the United States perform 175,000 to 500,000 procedures annually (2,3). Vasectomy is the fourth most common form of contraception, only behind condoms, oral contraceptives and tubal ligation (4). Despite improvements in the procedure and the development of the ‘No-scalpel’ or minimally-invasive technique, vasectomies continue to carry certain procedural risks including infection, symptomatic hematoma, vasectomy failure, and chronic post-operative pain, deemed post-vasectomy pain syndrome (PVPS). Intraoperative and early post-operative complications generally occur in 1 to 6% of vasectomies (5). Vasectomy, when compared to tubal ligation, however, is less likely to fail and less likely to result in post-operative complications (6).
Pain following vasectomy is not uncommon and can have a number of etiologies: hematoma, infection, nerve-impingement, hernia, testicular neoplasm and psychogenic causes. It is not until other potential causes have been ruled out that PVPS may be considered. That being said, there is no standardized definition for PVPS itself. Patients with PVPS may report orchialgia, which can be unilateral or bilateral, pain with intercourse, pain with activity, or tender/full epididymides (7).
The reported incidence of PVPS is highly variable. In 1992, McMahon et al. reported that 26 out of 172 of men surveyed (15%) reported chronic scrotal pain following vasectomy that they considered troublesome. A staggering 33% overall, however, reported chronic testicular discomfort following the procedure. Importantly, five percent of these men sought medical help and only two went on to pursue operative intervention (8). A similar retrospective postal questionnaire from Morris et al. in the United Kingdom showed that 6% of men sought medical advice for chronic scrotal pain following vasectomy (9). A prospective survey by Leslie et al. of men undergoing vasectomy from 2004–2006 returned with similar results. Of the 593 men who participated, 14.7% of men reported chronic scrotal pain at 7 months. Unfortunately, four of these men (0.9%) had pain severe enough to affect quality of life (10).
There are many studies indicating much lower rates of chronic pain, for instance, Leslie et al. demonstrated a rate of 0.9% of men reporting pain 7 months following the procedure (10). Furthermore, Choe et al. found at 4.8 years of follow-up only 2.2% of vasectomized men had pain severe enough to impact quality of life (11). In a comparative study, 6.0% of vasectomized men sought medical attention for chronic testicular pain compared to 2.0% of non-vasectomized men at 3.9 years of follow-up (9). For most men, it appears that the pain typically lasts from several weeks to months and generally dissipates prior to 1 year (5). Manikandan et al. reported that only 1.6% of men regretted having the procedure 10 years later due to pain (12). The American Urological Association recommends counseling men that chronic scrotal pain which negative impacts quality of life occurs in 1–2% of men following vasectomy (1).
From these studies, we are able to appreciate that PVPS is a painfully real entity that requires our attention. It is thought the pain may be a result of epididymal congestion, inflammatory factors such as interstitial or perineural fibrosis, vascular stasis, painful sperm granulomas, extravasation of sperm, anti-sperm antibody or entrapment of nerves at the operative site (7). Unfortunately, none of these hypotheses have been proven and the pathophysiology of PVPS remains elusive. A review of the literature on this subject shows many of these studies to be small, lacking in validated pain measures, consisting of poorly defined outcomes with short follow-up, and typically having high non-responder rates.
Conservative interventions for PVPS may include non-steroidal anti-inflammatory drugs (NSAIDs), tricyclic antidepressants (TCAs), local or regional nerve blocks, gabapentin, non-specific pain medications, acupuncture, and other complementary approaches. After these avenues have been exhausted, willing patients may consider operative intervention. The main surgical options for PVPS include vasectomy, epididymectomy and microsurgical spermatic cord denervation. In this review, we will focus on the evidence of vasectomy reversal as a means of alleviating PVPS.
Discussion
Vasectomy reversal seems to be the most logical surgical intervention for men who develop PVPS. If one relieves the obstruction then hopefully the pain will improve. Evidence of this may be postulated from Moss et al. who found a threefold increased relative risk of PVPS for closed-ended compared to open-ended vasectomy techniques (13). Pardanani observed epididymis with fullness, distention, turgidity, thickening, and induration in men undergoing undergoing vasovasostomy (14). Selikowitz reported in 1985 that epididymal specimens taken from men with unremitting epididymal pain following vasectomy demonstrated sequelae consistent with long-standing obstruction (15). Many of these changes are nearly universal in vasectomized men and therefore it remains unclear why this results in pain in some men, but not others (16).
There are several small series that would support claims that relief of the obstruction can improve pain in some men and absolve it in others. In an early study by Myers et al., 27 of 32 (84%) men with PVPS had resolution of pain following vasovasostomy. Of these 27 men, three men required a second vasectomy reversal to experience benefit. Not all of the reversals were true vasovasostomy, with vasoepididymostomy being required in 6 patients (17). A subsequent study performed, at the same institution, showed similar results. Of the thirty-one men that elected for vasectomy reversal, 82% of patients reported improvement in pain. There was a 59% improvement in pain scores in this group. Of note, two patients did require further intervention: one orchiectomy and one epididymectomy. Mean follow-up was 8.4 years, suggesting durability of response to the procedure for those who responded (18). Horovitz et al. were also able to look at long-term pain relief in a small group of men who pursued vasectomy reversal. Fourteen men were included in the study and the mean follow-up was 40.5±24.7 months. Following the procedure, 93% of the men reported improvement in pain and 50% reported being pain free. Unfortunately, 2 of the men (15%) reported recurrence of symptoms. Despite that, 93% of the men reported that they would undergo the same procedure again (19).
While most studies have looked only at surgical outcomes for vasectomy reversals, there are two that have tried to understand the disease process by looking at both histology and anatomy. Lee et al. looked at both general procedural satisfaction for vasectomy reversal and also at whether patency rates had any bearing on improvement of symptoms. This study showed a significant difference in improvement of pain scores when comparing the patent and non-patent group. The mean difference in pain scores was 6.00±1.25 (4-8) for the patent group and 4.43±0.98 (3-6) for the non-patent group (20). This may suggest that obstruction and congestion of the epididymis does indeed play a key role in the pathology. Using the excised tissue from patients undergoing vasectomy reversal as well as those undergoing initial vasectomy, Nangia et al. attempted to show histologic differences in those with PVPS. The group was looking specifically for degree of inflammation and nerve proliferation. In this particular group, 69% of the men who elected for vasectomy reversal became pain free. Unfortunately, there were no histological differences between the patients with PVPS or the matched controls. Nangia et al. also did not appreciate a histologic difference in those who reported improvement following vasectomy reversal and those who did not (7).
Despite a successful vasectomy reversal, however, some men will continue to have pain. As with all complex pain syndromes, psychological issues may contribute to the pain in men with chronic orchialgia. As evidence of this, Schover et al. evaluated men with genital pain and no organic findings. There were high rates of psychiatric disorders including somatization, nongenital chronic pain syndromes, major depression and chemical depression. Similarly, some of these men reported social isolation and had emotional loss at the time of pain onset (21). There remains a complex interplay of physical and psychiatric disorders in chronic pain syndromes with life stress, social support and other factors comingling. Men with preexisting chronic orchialgia should likely be steered away from vasectomy as a means of contraception.
Procedural choice is generally left to a discussion between the patient and provider, as aforementioned, epididymectomy or spermatic cord block with subsequent microsurgical cord denervation can be offered as well. That being said, there are perhaps important characteristics that can color this discussion. First and foremost, surgeon ability and training play an important role in what procedures are typically offered. This is most important for procedures that require microsurgical training including vasectomy reversal. Success rates for vasectomy reversal, regardless of when performed for pain or fertility are higher with a microsurgical approach and practitioners providing this service should be prepared and trained to perform the more complicated vasoepididymostomy if the situation arises. Table 1 provides an overview of success rates for vasectomy reversal in various studies. This may provide helpful information during the process of shared decision making.
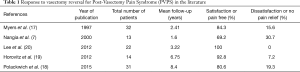
Full table
Another important factor to consider is possible restoration of fertility. While vasectomy reversal may have promising impact on pain control, providers must discuss the possibility of fertility. In fact, 36.4% of men undergoing vasectomy reversal went on to report viable pregnancies (20). With this in mind, Levine suggests epididymectomy for those patients with pain isolated solely to the epididymis or MDSC with diffuse pain. Vasectomy reversal is rarely offered (22).
Conclusions
Men with post-vasectomy pain should be evaluated to rule out other sources of discomfort. Conservative therapy with or without medical management is the appropriate initial treatment for most. How long to continue conservative treatment before proceeding to surgery is unclear. There are currently no guidelines or standardized protocols for which patients should proceed to surgical intervention. In the United States, vasectomy reversal generally remains an out-of-pocket expense and can carry significant financial burden which may delay or prevent its use entirely.
Following failure of more conservative therapies for PVPS, however, vasectomy reversal remains a reasonable treatment option. In the end, choice of surgery should be made after engaging in an in-depth discussion and using a patient-centered approach. Better studies are needed to characterize the incidence of PVPS according to standardized measures beginning shortly after the procedure and continued for long-term follow-up. In a similar fashion, larger studies equipped to evaluate the incidence of chronic pain and its varying severities and those patients reporting impaired quality of life, seeking medical help and receiving surgical procedures need to be better captured. Without better data, improvements in the diagnosis and treatment of PVPS will remain elusive.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Sharlip ID, Belker AM, Honig S, et al. Vasectomy: AUA guideline. J Urol 2012;188:2482-91. [Crossref] [PubMed]
- Barone MA, Hutchinson PL, Johnson CH, et al. Vasectomy in the United States, 2002. J Urol 2006;176:232-6; discussion 236. [Crossref] [PubMed]
- Eisenberg ML, Lipshultz LI. Estimating the number of vasectomies performed annually in the United States: data from the National Survey of Family Growth. J Urol 2010;184:2068-72. [Crossref] [PubMed]
- Martinez GM, Chandra A, Abma JC, et al. Fertility, contraception, and fatherhood: data on men and women from cycle 6 (2002) of the 2002 National Survey of Family Growth. Vital Health Stat 23 2006;26:1-142. [PubMed]
- Schwingl PJ, Guess HA. Safety and effectiveness of vasectomy. Fertil Steril 2000;73:923-36. [Crossref] [PubMed]
- Mosher WD, Martinez GM, Chandra A, et al. Use of contraception and use of family planning services in the United States: 1982-2002. Adv Data 2004;350:1-36. [PubMed]
- Nangia AK, Myles JL, Thomas Aj JR. Vasectomy reversal for the post-vasectomy pain syndrome: a clinical and histological evaluation. J Urol 2000;164:1939-42. [Crossref] [PubMed]
- McMahon AJ, Buckley J, Taylor A, et al. Chronic testicular pain following vasectomy. Br J Urol 1992;69:188-91. [Crossref] [PubMed]
- Morris C, Mishra K, Kirkman RJ. A study to assess the prevalence of chronic testicular pain in post-vasectomy men compared to non-vasectomised men. J Fam Plann Reprod Health Care 2002;28:142-4. [Crossref] [PubMed]
- Leslie TA, Illing RO, Cranston DW, et al. The incidence of chronic scrotal pain after vasectomy: a prospective audit. BJU Int 2007;100:1330-3. [Crossref] [PubMed]
- Choe JM, Kirkemo AK. Questionnaire-based outcomes study of nononcological post-vasectomy complications. J Urol 1996;155:1284-6. [Crossref] [PubMed]
- Manikandan R, Srirangam SJ, Pearson E, et al. Early and late morbidity after vasectomy: a comparison of chronic scrotal pain at 1 and 10 years. BJU Int 2004;93:571-4. [Crossref] [PubMed]
- Moss WM. A comparison of open-end versus closed-end vasectomies: a report on 6220 cases. Contraception 1992;46:521-5. [Crossref] [PubMed]
- Pardanani DS, Patil NG, Pawar HN. Some gross observations of the epididymides following vasectomy: a clinical study. Fertil Steril 1976;27:267-70. [Crossref] [PubMed]
- Selikowitz SM, Schned AR. A late post-vasectomy syndrome. J Urol 1985;134:494-7. [PubMed]
- Ishigami K, Abu-Yousef MM, El-Zein Y. Tubular ectasia of the epididymis: a sign of postvasectomy status. J Clin Ultrasound 2005;33:447-51. [Crossref] [PubMed]
- Myers SA, Mershon CE, Fuchs EF. Vasectomy reversal for treatment of the post-vasectomy pain syndrome. J Urol 1997;157:518-20. [Crossref] [PubMed]
- Polackwich AS, Tadros NN, Ostrowski KA, et al. Vasectomy reversal for postvasectomy pain syndrome: a study and literature review. Urology 2015;86:269-72. [Crossref] [PubMed]
- Horovitz D, Tjong V, Domes T, et al. Vasectomy reversal provides long-term pain relief for men with the post-vasectomy pain syndrome. J Urol 2012;187:613-7. [Crossref] [PubMed]
- Lee JY, Chang JS, Lee SH, et al. Efficacy of vasectomy reversal according to patency for the surgical treatment of postvasectomy pain syndrome. Int J Impot Res 2012;24:202-5. [Crossref] [PubMed]
- Schover LR. Psychological factors in men with genital pain. Cleve Clin J Med 1990;57:697-700. [Crossref] [PubMed]
- Tan WP, Levine LA. An overview of the management of post-vasectomy pain syndrome. Asian J Androl 2016;18:332-7. [Crossref] [PubMed]


