Laparoscopic adrenalectomy for pheochromocytoma-does size matter? A single surgeon comparative study
Introduction
Catecholamine-secreting tumors arise from neuroectodermal tissue of the neural crest and are called pheochromocytomas when they arise in the adrenal gland and paragangliomas when the origin lies outside the adrenal (1). Surgical removal is the primary management modality and minimally invasive surgery is the current standard of care (1). Pheochromocytomas contain large amounts of catecholamines and catecholamine surges during tumor manipulation may exceed normal values by 1,000–1,500 times (2,3). Therefore, one of the major concerns with this surgery are the intra-operative hemodynamic changes which can result in adverse cardio-vascular events including pulmonary edema, myocardial infarction and stroke. Pre-operative patient preparation contributes to a safe surgery but it is also important that the surgery be quick and with minimal tumor handling (2). Increasing tumor size can result in greater operative difficulty which may translate into higher intra-operative complications and conversion and there is debate about the use of laparoscopy for large tumors (4,5). We retrospectively reviewed our data of laparoscopic surgery for pheochromocytomas and compared the outcomes between patients with large (>4 cm) and small tumors.
Methods
A retrospective review was performed of all patients who underwent laparoscopic adrenalectomy for pheochromocytomas by a single surgeon between July 2010 and August 2013. Case-sheets were retrieved and reviewed for surgical outcomes. Data for patients with large tumors (>4 cm) was compared with the cohort with small tumors. All patients with bilateral tumors had one tumor >4 cm while the other tumor was smaller. Operative data for these patients was equally divided between the two groups. Non-parametric statistical tests (Mann Whitney U test) were used for comparison of data.
The diagnosis of a catecholamine secreting tumor was based on standard clinical criteria including symptoms, hormone evaluation, cross-sectional imaging (contrast enhanced computed tomography or MRI) and functional imaging with (123I)-labeled metaiodobenzylguanidine scintigraphy or 68Ga-labelled (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid)-1-NaI[3]-Octreotide whole body positron emission tomography-computed tomography where indicated (6).
All patients underwent preoperative stabilization with a combination of α and β adrenergic blockers and volume expansion. All patients received general anaesthesia with endotracheal intubation. A transperitoneal approach was used in all patients. With the patient in a lateral decubitus position, three ports were used for most cases with an additional 4th port for liver retraction on the right side. All specimens were entrapped in a specimen bag and most were retrieved through a muscle splitting iliac fossa incision. Very small specimens were retrieved, with the bag, through a 10 mm port site. A drain was placed in all cases and was removed if 24-hour output was under 30 mL. Patients were allowed orally the same evening and discharged after drain removal. All patients provided written informed consent for the procedures.
Results
Twenty four patients underwent 28 procedures during the period under review. Ten patients were female. The overall age range was 13–62 years (mean 35 years). Fifteen tumors were greater than 4 cm in size. Four patients had bilateral tumors where one tumor in each patient was greater than 4 cm. Among the remaining 20 patients, 9 had unilateral small tumors and 11 had unilateral large tumors. The tumor size ranged from 0.7 to 4 cm (mean 2.95 cm) in the small tumor group and from 4.1 to 9.5 cm (mean 6.3 cm) in the large tumor group. Three tumors were greater than 8 cm in size.
Patients with larger tumors had similar operating time (138 vs. 117 min; P=0.2) and blood loss (181 vs. 143 mL; P=0.41) as patients in the small tumor group. No patient required blood transfusions; there were no intra-operative complications or conversions to open surgery. The small tumor group had four Clavien-Dindo (7) grade 1 and one Clavien-Dindo grade 3a complications while the large tumor group had three Clavien-Dindo grade 1 complications. Eighteen patients (75%) did not require any anti-hypertensive medications post-operatively while 6 of 24 required lower doses (3 with small tumors, 2 with large tumors, and 1 with bilateral tumors). The grade 3a complication was a sub-hepatic hematoma that was managed with ultrasound guided drainage. Table 1 summarizes the data. All tumors were benign on histology.
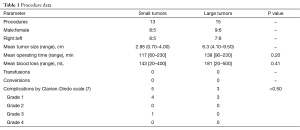
Full table
Discussion
Pheochromocytoma surgery has generally been considered a relatively high-risk procedure due to the hemodynamic changes that occur during and after surgery (8,9). There has been some debate about using laparoscopy, particularly for larger tumors, because of concerns regarding pneumoperitoneum, tumor manipulation, and prolonged surgery duration, all of which potentially increase catecholamine release (4,5,10,11). However, laparoscopy is recognized as the standard of care management for most pheochromocytomas and pre-operative preparation for up to 2 weeks with adrenoceptor blockade and volume expansion results in a decrease in intraoperative risks and complications (2,12). Studies comparing open with laparoscopic surgery attest to the safety of the laparoscopic approach, irrespective of tumor size (13).
Our operative data for small and large tumors shows no significant difference. Operative times, blood loss and complications were similar in the two groups even though the mean tumor size was more than 2 times greater in the large tumor group. These findings are similar to those previously reported and support the belief that size alone may not be a criterion for selecting the surgical approach (4,14). Team development and surgeon experience have a significant role to play in decreasing operative time and complications in all advanced surgical procedures (13). This would be particularly relevant in conditions such as pheochromocytomas where a multidisciplinary involvement is required at all times Once a specific surgical technique has been established, tumor size alone is unlikely to impact outcomes until it reaches proportions that significantly distort anatomy or limit access.
Large tumors may be associated with a greater number of intraoperative hemodynamic events as the catecholamine content within tumors may be directly proportional to their size and tumor manipulation is an important stimulus for their release (15-17). However, we did not encounter any increase in adverse vascular events. This is consistent with previous reports, including ours, where such complications were absent despite pressure surges above 300 mmHg in some patients (5,18,19). The two major reasons for this lack of adverse outcomes are the short duration of such surges and the improvements in anesthetic techniques and drugs which can rapidly control them. In addition, surgeon expertise decreases the number of instances of direct tumor manipulation and operative time, lowering the incidence of complications (4).
Our data is limited by the fact that it is retrospective in nature and there are potential differences in the preparation and management protocols of the patients. Prospective studies controlling for such variables would allow a more robust evaluation of these phenomenon.
Conclusions
Laparoscopic adrenalectomy is safe and effective for managing pheochromocytomas, irrespective of tumor size. Surgery for larger tumors is associated with no increase in adverse outcomes.
Acknowledgements
None.
Footnote
Conflicts of Interest: The paper has previously been presented at the Asian Congress of Urology and conference abstracts were published as R Kumar, N Rao, R Ramachandran, P Singh, N Tandon. Laparoscopic adrenalectomy for pheochromocytoma: comparative outcome analysis of small versus large tumors. Int J Urol 2014;19:S114.
Ethical Statement: Written informed consent was obtained from all patients prior to surgery.
References
- Lenders JW, Eisenhofer G, Mannelli M, et al. Phaeochromocytoma. Lancet 2005;366:665-75. [Crossref] [PubMed]
- Pacak K. Preoperative management of the pheochromocytoma patient. J Clin Endocrinol Metab 2007;92:4069-79. [Crossref] [PubMed]
- Schüttler J, Westhofen P, Kania U, et al. Quantitative assessment of catecholamine secretion as a rational principle of anesthesia management in pheochromocytoma surgery. Anasthesiol Intensivmed Notfallmed Schmerzther 1995;30:341-9. [PubMed]
- Carter YM, Mazeh H, Sippel RS, et al. Safety and feasibility of laparoscopic resection for large (≥ 6 CM) pheochromocytomas without suspected malignancy. Endocr Pract 2012;18:720-6. [Crossref] [PubMed]
- Conzo G, Pasquali D, Della Pietra C, et al. Laparoscopic adrenal surgery: ten-year experience in a single institution. BMC Surg 2013;13 Suppl 2:S5. [Crossref] [PubMed]
- Naswa N, Sharma P, Nazar AH, et al. Prospective evaluation of 68Ga-DOTA-NOC PET-CT in phaeochromocytoma and paraganglioma: preliminary results from a single centre study. Eur Radiol 2012;22:710-9. [Crossref] [PubMed]
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13. [Crossref] [PubMed]
- Tauzin-Fin P, Hilbert G, Krol-Houdek M, et al. Mydriasis and acute pulmonary oedema complicating laparoscopic removal of phaechromocytoma. Anaesth Intensive Care 1999;27:646-9. [PubMed]
- Quezado ZN, Keiser HR, Parker MM. Reversible myocardial depression after massive catecholamine release from a pheochromocytoma. Crit Care Med 1992;20:549-51. [Crossref] [PubMed]
- Joris JL, Hamoir EE, Hartstein GM, et al. Hemodynamic changes and catecholamine release during laparoscopic adrenalectomy for pheochromocytoma. Anesth Analg 1999;88:16-21. [PubMed]
- Kinney MA, Warner ME, vanHeerden JA, et al. Perianesthetic risks and outcomes of pheochromocytoma and paraganglioma resection. Anesth Analg 2000;91:1118-23. [PubMed]
- Chen H, Sippel RS, O'Dorisio MS, et al. The North American Neuroendocrine Tumor Society consensus guideline for the diagnosis and management of neuroendocrine tumors: pheochromocytoma, paraganglioma, and medullary thyroid cancer. Pancreas 2010;39:775-83. [Crossref] [PubMed]
- Lodin M, Privitera A, Giannone G. Laparoscopic adrenalectomy (LA): keys to success: correct surgical indications, adequate preoperative preparation, surgical team experience. Surg Laparosc Endosc Percutan Tech 2007;17:392-5. [Crossref] [PubMed]
- Toniato A, Boschin IM, Opocher G, et al. Is the laparoscopic adrenalectomy for pheochromocytoma the best treatment? Surgery 2007;141:723-7. [Crossref] [PubMed]
- Huynh TT, Pacak K, Brouwers FM, et al. Different expression of catecholamine transporters in phaeochromocytomas from patients with von Hippel-Lindau syndrome and multiple endocrine neoplasia type 2. Eur J Endocrinol 2005;153:551-63. [Crossref] [PubMed]
- Eisenhofer G, Lenders JW, Goldstein DS, et al. Pheochromocytoma catecholamine phenotypes and prediction of tumor size and location by use of plasma free metanephrines. Clin Chem 2005;51:735-44. [Crossref] [PubMed]
- Flávio Rocha M, Faramarzi-Roques R, Tauzin-Fin P, et al. Laparoscopic surgery for pheochromocytoma. Eur Urol 2004;45:226-32. [Crossref] [PubMed]
- Tauzin-Fin P, Sesay M, Gosse P, et al. Effects of perioperative alpha1 block on haemodynamic control during laparoscopic surgery for phaeochromocytoma. Br J Anaesth 2004;92:512-7. [Crossref] [PubMed]
- Ramachandran R, Rewari V, Sharma A, et al. Laparoscopic Surgery for Pheochromocytoma and Paraganglioma Removal: A Retrospective Analysis of Anaesthetic Management. Curr Hypertens Rev 2016. [Epub ahead of print]. [Crossref] [PubMed]


